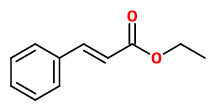
Vanilla planifolia Andrews - syn.Vanilla fragrans Ames; Vanilla aromatica Willd. - Orchidaceae
vanilla, Mexican vanilla, Vera Cruz vanilla, bourbon vanilla, (Echte) Vanille
Evergreen vine, native to Central America, cultivated elsewhere (Madagascar); flowers greenish-yellow;
„The fruits are 15-23 cm (6-9 in) long pods (often incorrectly called beans)… Vanillin was first isolated as a relatively pure substance in 1858 by Nicolas-Theodore Gobley, who obtained it by evaporating a vanilla extract to dryness, and recrystallizing the resulting solids from hot water.“
http://en.wikipedia.org/wiki/Vanilla_planifolia
[Gobley, N.-T. (1858). „Recherches sur le principe odorant de la vanille“. Journal de Pharmacie et de Chimie. 34: 401-405]
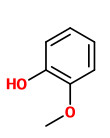
Quantitative analysis (GC) of vanilla oleoresin revealed 1 ppm of cis/trans-vitispirane in the ratio of 1:3. The cis diastereomer seems fresher and more intense (reminescent of the green scent of chrysanthemum, with a flowery-fruity wine note) than the trans-compound (heavy exotic flower scent with an earthy-woody touch).
[Schulte‐Elte, Karl H., et al. „Vitispiranes, Important Constitutents of Vanilla Aroma.“ Helvetica Chimica Acta 61.3 (1978): 1125-1133]
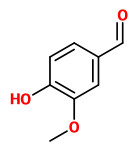
The extract of green vanilla beans contained glucosides of vanillin, vanillic acid, p-hydroxy benzaldehyde, vanillyl alcohol, p-cresol, and creosol.
[Dignum, Mark JW, et al. „Identification of glucosides in green beans of Vanilla planifolia Andrews and kinetics of vanilla β-glucosidase.“ Food Chemistry 85.2 (2004): 199-205]
„The level of the aldehydes, vanillin and p-hydroxybenzaldehyde and their respective acids (vanillic acid and p-hydroxybenzoic acid), in cured vanilla beans is used as an indicator of cured vanilla bean quality for commercial purposes. Although they are important, these compounds alone do not account for the flavour strengthand extremely complex structure of this flavouring. Ten phenolic compounds were detected in vanilla extracts as being aroma-active.Guaiacol, 4-methylguaiacol and acetovanillone, occurring at concentrations of 3.8-13.7 ppm, were similar in intensity to vanillin, which was detected at a concentration of more than 1000 times that of these compounds. Methyl salicylate, detected at a level of less than 1 ppm, was perceived as being as intense as vanillin. p-Cresol, methyl cinnamate and anisyl alcohol, occurring at concentrations of 1.1-2.4 ppm, were of medium intensity. Sweet, woody, balsamic, spicy, vanilla-like and toasted notes were attributed to phenolic compounds. Vanillic acid was not perceived by panellists, because its elution required a high temperature, which caused a burnt odour in the sniffing port. The aldehydes 2-heptenal and (E)-2-decenal, identified here for the first time in vanilla beans, were perceived as being of medium intensity, with green, oily and herb-like floral notes. Aliphatic, acetic, isobutyric, isovaleric and valeric acids were perceived by the panellists as having sour, buttery and oily notes. In conclusion, this study is in agreement with previous observations of the contribution of minor constituents to the overall aroma of cured vanilla beans, through GC-O analysis of a representative aroma extract from cured vanilla beans.“
[GC-MS and GC-olfactometry analysis of aroma compounds in a representative organic aroma extract from cured vanilla (Vanilla planifolia G. Jackson) beans., Pérez-Silva, A., Odoux, E., Brat, P., Ribeyre, F., Rodriguez-Jimenes, G., Robles-Olvera, V., Günata, Z., Food chemistry, Vol.99(4), 2006, 728-735]
By GC-MS and GC-O, 78 odorants were identified in traditionally cured Bourbon and Ungandan vanilla bean extracts. Vanillin was the most abundant followed by guaiacol (105/169ppm) and the two isomers of 2,3-butandiol (36/46ppm; plus their acetals with vanillin 30/14ppm). In the range of 1-10ppm were 3-methyl-2-butanone, acetoin, isovaleric acid, γ-butyrolactone, phenol, pantolactone, benzyl alcohol, 4-methylphenol, octanol, phenylethanol, benzoic acid, octanoic acid, 4-methylguaiacol, methyl salicylate, p-anisaldehyde, nonanoic acid, anisyl alcohol, cinnamyl alcohol, isovanillin, cinnamic acid, ethyl cinnamate, acetovanillone, methyl vanillate, cis-18-heptacosene-2,4-dione, and cis-20-nonacosene-2,4-dione. Carvacrol and β-damascenone were only found in Ugandan beans. Among the twenty-three most powerful odorants (medium or strong odor intensity, but ⇐ 0.05ppm) were 3,5-octadien-2-one, nonanol, γ-octalactone, 4-allylphenol, heliotropine, methyl decanoate, β-damascenone, and some nitrogen-containing compounds. „Interestingly, of the six more concentrated strong odorants, namely, acetic acid, methyl salicylate, p-anisaldehyde, methyl trans-cinnamate, trans-cinnamic acid, and methylparaben, five were more abundant in the Ugandan vanilla beans, with p-anisaldehyde as the most different in terms of concentration.“
[Zhang, Suying, and Christoph Mueller. „Comparative analysis of volatiles in traditionally cured Bourbon and Ugandan vanilla bean (Vanilla planifolia) extracts.“ Journal of agricultural and food chemistry 60.42 (2012): 10433-10444]
Correlating well with previously reported data from the literature, the „good“ vanilla samples contained high concentrations of acetic acid, 2-methoxy phenol, hydroxyl dihydromaltol, 5-(hydroxymethyl) furan-2-carbaldehyde, 4-hydroxybenzaldehyde, hexadecanoic acid, and 1-octadecanol.
„Bad“ vanilla beans were noticed with high concentrations of 2-methoxy phenol, 2-methoxy-4-methyl-phenol, hexadecanoic acid, and 1-octadecanol, as well as low concentrations of vanillin, hydroxyl dihydro maltol and hydroxymethyl furfural.
[S.J. Toth, Comparison and integration of analytical methods for the characterization of vanilla chemistry. Proquest, Uni Dissertation Publishing (2012)]
„…the most odor-active compounds (FD 3125), vanillin (sweet), guaiacol (phenolic), ethyl (E)-cinnamate (fruity) and β-damascenone (raisin-like), were common to red whole and cuts beans. Moreover, in the same FD factor range (FD 3125), 2-methylbutanoic acid (buttery), 3-methylbutanoic acid (buttery) and p-cresol (fecal) contributed more to the aroma of MV red whole, whereas γ-nonalactone (coconut-like) and eugenol (clove-like) contributed more to the aroma of MV cuts. Based on their odor qualities, vanillin and guaiacol respectively contributed to the sweet and phenolic attributes by the sensory evaluation, and both ethyl (E)-cinnamate and β-damascenone contributed to the resinous and dried fruit-like attributes.“
[Takahashi, Makoto, et al. „Key odorants in cured Madagascar vanilla beans (Vanilla planiforia) of differing bean quality.“ Bioscience, biotechnology, and biochemistry 77.3 (2013): 606-611] https://www.jstage.jst.go.jp/article/bbb/77/3/77_120842/_pdf
Quantitative analysis (HPLC) of vanilla extracts showed mean concentrations of guaiacol and vanillin (µg/mL) in 'MV red whole' with standard quality (17.8/2480) and 'MV cuts' with substandard aroma (88.7/1540).
[Takahashi, Makoto, Shizuka Sakamaki, and Akira Fujita. „Simultaneous analysis of guaiacol and vanillin in a vanilla extract by using high-performance liquid chromatography with electrochemical detection.“ Bioscience, biotechnology, and biochemistry 77.3 (2013): 595-600] https://www.jstage.jst.go.jp/article/bbb/77/3/77_120835/_pdf
„There are different methods of curing, and each one is unique and named after the places of its origin like Mexican process and Bourbon process. Recently, Central Food Technological Research Institute, Mysore has developed know-how of improved curing process, where the green vanilla beans are cured immediately after harvest and this process takes only 32 days, which otherwise requires minimum of 150-180 days as reported in traditional curing methods.“
[Vanilla-Its Science of Cultivation, Curing, Chemistry, and Nutraceutical Properties., Anuradha, K., Shyamala, B.N., Naidu, M.M., Critical reviews in food science and nutrition, Vol.53(12), 2013, 1250-1276]
„HS-SPME combined with GC-MS was used to analyze the volatile components from V.planifolia fruits… A total of 28 components were separated and identified, accounted for 98.17% of total volatile components. Among them, vanillin had the maximum content (48.28%), followed with guaiacol (15.54%).“
[Analysis of the volatile components in the fruits of Vanilla planifolia Andrews by HS-SPME combined with GC-MS., Huan, L., JinQing, L., Yi, D., XiaoShuang, L., ShengNan, G., Qiang, L., Medicinal Plant, Vol.5(4), 2014, 23-26]
Main volatile compounds of SDE extracts from Bourbon vanilla from Madagascar were furfural, phenol, guaiacol, p-vinylguaiacol and vanillin.
„Vanilla planifolia showed stronger phenolic, woody, smoky notes [than V. tahitensis] due to guaiacol, creosol and phenol, which were found to be biomarkers of the species. Vanilla sensory properties were linked by partial least squares regression to key volatile compounds like guaiacol or creosol, which are indicators of lower quality.“
[Brunschwig, Christel, et al. „Volatile composition and sensory properties of Vanilla× tahitensis bring new insights for vanilla quality control.“ Journal of the Science of Food and Agriculture 96.3 (2016): 848-858]
„Though cis-6-dodecen-4-olide [dairy lactone] has a similar chemical structure as gamma dodecalactone, its threshold value is 700 times lower than that of gamma dodecalactone and it affects the vanilla beans aroma only with a slight amount. A plenty of experiences and keen sense of smell of our researchers discovered the contribution of a trace of this component… cis-6-Dodecen-4-olide enhances the whole vanilla note and lasting sweetness when added to vanilla flavor at a certain concentration.“
http://www.soda-sales.com/15054409387349 Retrieved 21 June 2018.
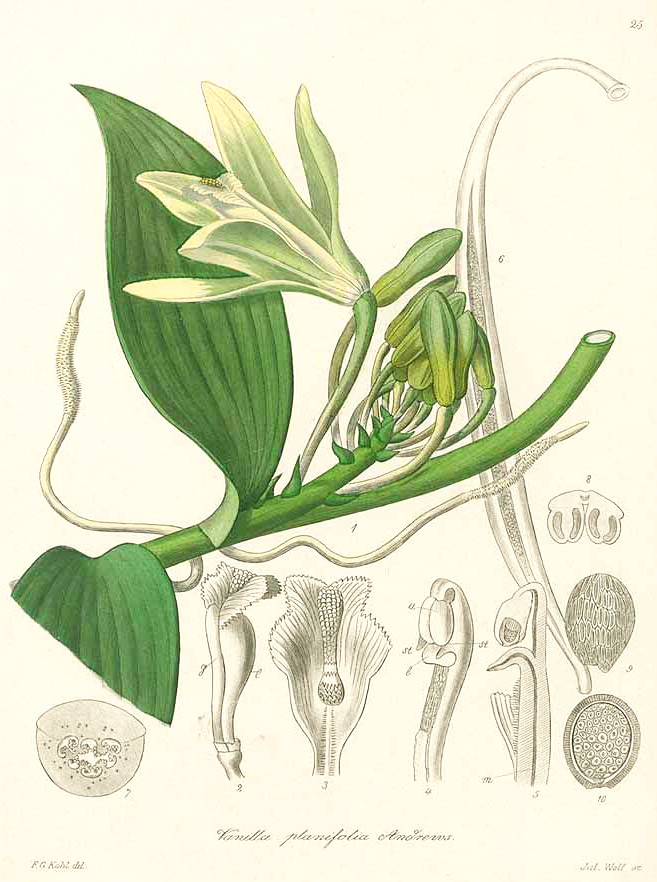
Kohl, F.G., Die officinellen Pflanzen der Pharmacopoea Germanica, t. 25 (1891-1895) [F.G. Kohl]
http://www.plantillustrations.org/species.php?id_species=1051111

Vanilla planifolia flowering in Florida Southern College's greenhouses (2012)
CC BY-SA 2.0
Author: Malcolm Manners from Lakeland FL, USA Wikimedia Commons