Tilia cordata Mill. - syn. Tilia parvifolia Ehrh. - Malvaceae
little-leaf linden, small-leaved lime, Winterlinde, Kleinblättrige Linde, Steinlinde
Native tree to Europe and West Sibiria, up to 30m high; leaves with darker green upper side and blueish green underside, beneath glaucous; flowers with pleasant scent. There are numerous hybrids planted: Tilia x europaea = Tilia x vulgaris = Tilia cordata x Tilia platyphyllos
Dried flowers ared used as diaphoretic, as described for Tilia platyphyllos.
„In the countries of Central and Western Europe, linden flowers are a traditional herbal remedy made into an herbal…, considered to be of value as an anti-inflammatory in a range of respiratory problems: colds, fever, flu, sore throat, bronchitis, and cough.“ http://en.wikipedia.org/wiki/Tilia_cordata
„Thus the difference in the composition of the aroma compounds between the headspace and the essential oil is clearly shown. This is also the case with dried lime tree flowers (Flores Tiliae). The main constituents of the essential oil of this drug are 1,8-cineole, linalol, carvone, thymol and carvacrol (concentration more than 2 %), but all of these volatiles only be detected in the headspace in a concentration below. On the other hand p-cymene, fenchone, and α- and β-thujone are only trace compounds of this essential oil.
As main constituents of the headspace of lime tree flowers (Flores Tiliae), p-cymene, fenchone, α-thujone, β-thujone, camphor, anethole and menthone (each above 5 %) were found. In addition, more than forty other compounds, mainly terpenes, were identified. Also in this headspace sample a lot of aromatic compounds were found, which may have an important influence on the odour impression of Flores Tiliae. The olfactometric investigation of the headspace of the dried lime tree flowers by the GC-sniffing technique showed also in this case, that no compound on its own was responsible for the typical odour of these flowers. The essential oil from lime tree flowers can be described as follows: spicy, reminiscent of lovage, woody and also reminiscent of the dry part and of the animal part of neroli.“
[Headspace Analysis of the Dried Herb of Passion Flower (Herba Passiflorae) and Dried Flowers of Lime Tree (Flores Tiliae), G. Buchbauer, J. Jirovetz, B. Remberg, G. Remberg and A. Nikiforov, Flav. Frag. J., 7(6), 329 (1992)]
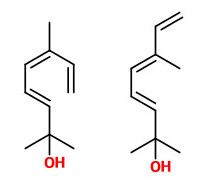 2,6-dimethyl-3(E),5(Z/E),7-octatriene-2-ol (sweet floral)
2,6-dimethyl-3(E),5(Z/E),7-octatriene-2-ol (sweet floral)
The highly sensitive 2,6-dimethyl-3(E),5(Z),7-octatriene-2-ol and its 5(E) isomer (natural ratio 1:10, sweet floral odor) are major constituents present only in the headspace and not in extracts of the flowers of T.cordata.
[Trapping, Investigation and Reconstitution of Flower Scents, Roman Kaiser, in: Müller, P.M., and Lamparsky, D. eds. Perfumes: Art, Science and Technology. Springer Science & Business Media, Dordrecht 1994, 213-250] but
Later on, headspace analysis of hyacinth flowers showed that 2,6-dimethylocta-1,3,5,7-tetraenes and 2,6-dimethylocta-3,5,7-triene-2-ols are associated with charcoal as adsorbing material. It was shown that these compounds are artefacts from ocimene which mainly arise during sampling with charcoal.
[Headspace analysis of hyacinth flowers. Brunke, E. J., Hammerschmidt, F. J., Schmaus, G., Flavour and fragrance journal, Vol.9(2), 1994, 59-69]
„More than 45 headspace constituents of the living lime tree flowers were detected by gas chromatographic-spectroscopic methods; more than 35 of them were identified. Main components (concentration higher than 3%) of this mixture with an odour impression similar to that of the genuine lime tree flowers' as well as with weak fresh and aromatic side-notes were limonene (22%), p-cymene (22%), δ3-carene (15%), germacrene-D (9%), β-phellandrene (4%) and farnesol (4%). Using a GC-sniffing technique, regions of specific odour impressions were found by means of which the total odour of the headspace sample of the living flowers was determined: the aromatic odour was caused especially by p-cymene, trans-carveol and germacrene-D; the floral part by linalool, 2-phenylethanol, trans-rose oxide, geraniol, nerol, 2-phenylethyl acetate, geranyl acetate and partly, nerolidol; the fresh part by camphene, limonene and camphor; and the narcotic part by 2-phenylethanol, nerol, heliotropin and farnesol. However, indole, 2-phenylethyl benzoate, farnesyl acetate and some other sesquiterpenes had additional odour notes…
The heavy sweetness and sultriness of the fragrance of the lime tree flowers undoubtedly was due to the content of indole and 2-phenylethyl acetate.“
[Comprative headspace anaysis of living and fresh cut lime tree flowers (Tiliae flores). G. Buchbauer, B. Remberg, L. Jirovetz, A. Nikiforov, Flavour and Fragrance Journal (1995) 10(3):221 - 224]
In comparatively small amounts, unsaturated C9 aldehydes and alcohols (catabolites of linolenic acid) are important scent modifiers in violet flowers, linden blossoms and Acacia farnesiana flowers.
(Add.: 0.02% (E,Z)-2,6-Nonadienal was found in the headspace of linden flowers; an extract of the flowers contained also (Z)-3-nonen-1-ol, (E)-2-nonen-1-ol, (Z)-6-nonen-1-ol, (Z,Z)-3,6-nonadien-1-ol, and (E,Z)-2,6-nonadienol; R.Kaiser pers.comm.)
[Meaningful Scents around the World, Roman Kaiser, Zürich 2006, 43]
„The essential oils of linden blossom from the clean localities contained 47.3-55.0% of oxygenated constituents, while the blossom oils from trees growing on the tile-covered pavements contained only 20.6-27.7% of these compounds. The major constituents determined in linden blossom oils were 3-p-menthene, nonanal, nonanoic acid, α-cadinene, hexahydrofarnesyl acetone, kaurene, aliphatic hydrocarbons (C20H42 - C24H50). The identified compounds (169 out of 250) made up 73.4-97.7% of the oils.“
[Influence of urban environment on chemical composition of Tilia cordata essential oil. Nivinskienė, Ona; Butkienė, Rita; Gudalevič, Alena; Mockutė, Danutė; Meškauskienė, Vilma; Grigaliūnaitė, Banga, Chemija, 2007, Vol. 18, No. 1, 44-49]
As shown by aroma extract dilution analysis (AEDA), linden honey volatiles having high factors of dilution (FD) were identified as 1-hexen-3-one, 2-acetyl-1-pyrroline, dimethyl trisulphide, methional, phenylacetaldehyde, 2-phenylethanol, linalool, p-cresol, 3,9-epoxy-1-p-menthene, 4-methylacetophenone, 3,9-epoxy-1,4(8)-p-menthadiene (linden ether), 1,3-p-menthadien-7-al, p-anisaldehyde, 4-vinylguaiacol, (E)-β-damascenone, eugenol, vanillin and cis-rose oxide.
[Intensive neutral odourants of linden honey Differences from honeys of other botanical origin., Blank, I., Fischer, K.H., Grosch, W., Zeitschrift für Lebensmittel-Untersuchung und Forschung, 189(5), 1989, 426-433] http://imreblank.ch/ZLUF_1989_189_426.pdf
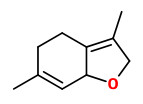 linden ether (floral mint) | 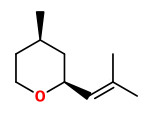 cis-rose oxide (floral green) |
„It was shown that 3.9-epoxy-1.4(8)-p-menthadiene and cis-rose oxide, both occurring also in the blossoms of the lime tree Tilia cordata (L.), were typical odourants of the linden honey.“
[Determination of the Chemical Structure of Linden Ether. Imre Blank, Werner Grosch, Wolfgang Eisenreieh, Adelbert Bacher, and Joachim Firl, Helvetica Chimica Acta, Volume 73 (1990), 1250-1257] http://imreblank.ch/HCA_1990_73_1250.pdf
Linden nectar extracts contained compounds of fatty acid degradation (nonanal, decanal, tetradec-1-ene), phenylpropanoids (3-(4-methoxyphenyl)propan-1-ol, 3-(4-methoxyphenyl)propanal, 3-(4-methoxyphenyl)prop-2-enal), isoprenoids (vomifolione, vomifoliol, and 3,5,5-trimethyl-4-(3-oxobutyl)cyclohex-2-en-1-one), alkaloids (caffeine, theophylline, a trace of nicotine), and a complex mixture of monoterpenes, among them linden ether and 1,8-cineole. Main components of ripe linden bee honey were benzoic acid and phenylacetic acid, together with two new monoterpenic acids, 4-isopropenylcyclohexa-1,3-diene-1-carboxylic acid and 4-(1-hydroxy-1-methyl-ethyl)cyclohexa-1,3-diene-1-carboxylic acid.
[Naef, Regula, et al. „From the linden flower to linden honey-volatile constituents of linden nectar, the extract of bee‐stomach and ripe honey.“ Perspectives in Flavor and Fragrance Research (2005): 31-40] http://www.untiredwithloving.org/honey_bee_linden.pdf
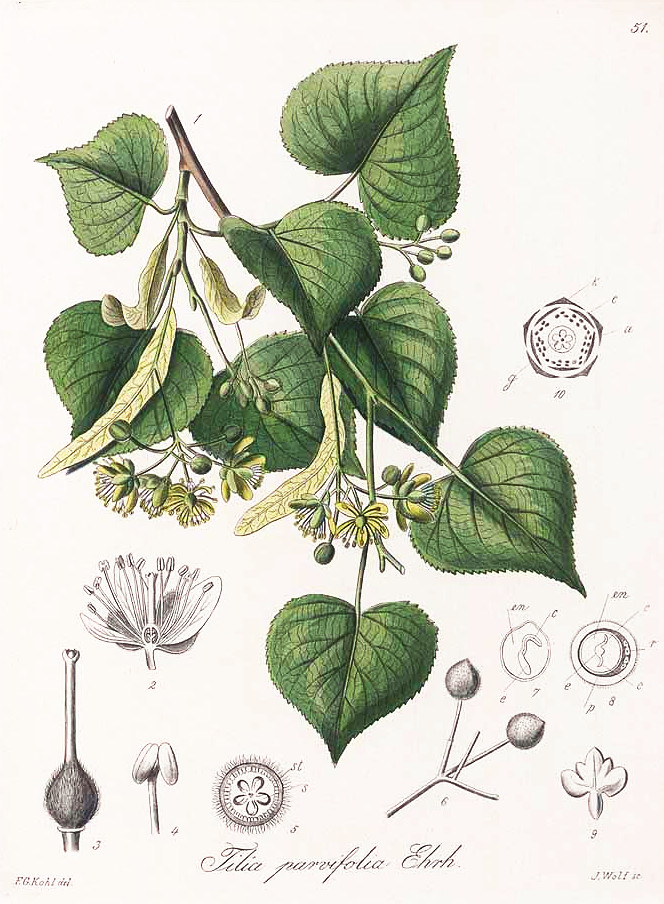
Tilia cordata as Tilia parviflora: Kohl, F.G., Die officinellen Pflanzen der Pharmacopoea Germanica, t.51 (1891-1895)
http://plantgenera.org/species.php?id_species=1017551

Tilia cordata flowers, CC BY-SA 3.0, Author: Andreas Kraska
