Abies balsamea (L.) Mill. - syn.Pinus balsamea L. - Pinaceae - balsam fir, Canada balsam, eastern fir, Balsamtanne
Tree, native to northeastern north America; leaves fragrant, 1-2.5cm long, 1-ranked to spiraled; pollen cones at pollination red, purplish, bluish, greenish, or orange; seed cones cylindric, gray-purple, turning brown before scale shed. efloras.org
„Canada balsam, also called Canada turpentine or balsam of fir, is a turpentine which is made from the resin of the balsam fir tree (Abies balsamea) of boreal North America. The resin, dissolved in essential oils, is a viscous, sticky, colourless or yellowish liquid that turns to a transparent yellowish mass when the essential oils have been allowed to evaporate… Due to its high optical quality and the similarity of its refractive index to that of crown glass (n=1.55), purified and filtered Canada balsam was traditionally used in optics as an invisible-when-dry glue for glass, such as lens elements… Canada balsam was also commonly used for making permanent microscope slides.“ wikipedia
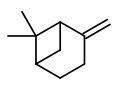
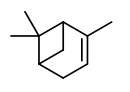
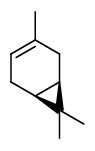
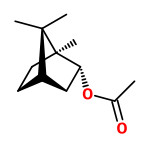
„Canada balsam comprises about 55% of diterpenoid acids, mainly abietic, neoabietic and palustric acids, 26% of volatile terpene hydrocarbons and about 19% of neutral diterpenoids of which the larger part consists of abienol.“
[Gray, P. S., and J. S. Mills. „1109. The isolation of abienol from Canada Balsam, the oleoresin of Abies balsamea (L.) Mill.“ Journal of the Chemical Society (Resumed) (1964): 5822-5825]
Steam distillation of foliage from Abies balsamea yielded 0.59% of an essential oil. Components were β-pinene (36%), bornyl acetate (14%), Δ3-carene (11%), limonene (11%), α-pinene (8%), camphene (6%), myrcene (2%), santene (1%), tricyclene (1%) and camphor, borneol, d-canadene, bisabolene, and terpinolene (all below 1%).
[The essential oil of Abies balsamea (L.) Mill., Shaw, A.C., Canadian Journal of Chemistry, Vol.31(2), 1953, 193-199] PDF
„Monoterpenoids and sesquiterpene hydrocarbons of Abies balsamea cortical oleoresin (Canada balsam) were analyzed by a combination of chromatographic and spectroscopic methods. Monoterpene hydrocarbons (21%) were composed of β-pinene, α-pinene, β-phellandrene, limonene, 3-carene, myrcene and camphene (listed in order of decreasing percentages), and oxygenated monoterpenes (0·4%) contained 4,4-dimethyl-2-cyclohepten-1-one, linalool, bornyl acetate, methylthymol, citronellyl acetate, α-terpineol, piperitone, citronellal, borneol, citronellol, two unknowns, and geraniol. From the sesquiterpene hydrocarbon fraction (1·1%) were isolated: longifolene, β-bisabolene, longipinene, an unknown, sativene, cyclosativene, cis-α-bisabolene, β-himachalene, α-himachalene, β-caryophyllene, γ-humulene, farnesene, longicyclene, an unknown, and β-selinene. Both himachalenes have been identified for the first time in Pinaceae outside of Cedrus; their co-occurrence with γ-humulene, longifolene, longipinene and longicyclene supports the biosynthetic mechanism by which all of these compounds arise through initial 1/11 cyclization of tran-cis-farnesylphosphate.“
[Chemical composition of the cortical essential oil from Abies balsamea., Chung, J.L., Snajberk, K., Zavarin, E., Phytochemistry, Vol.13(1), 1974, 179-183]
„The composition of the essential oils of 29 individual trees of Abies balsamea (L.) Mill, were investigated by a combination of retention times (GC with capillary columns) and GC/MS. The oils were obtained by steam distillation from various parts of plants harvested north of Forestville, Quebec. A total of 209 peaks were detected by chromatography of which a maximum of 106 peaks were recorded for one sample. The composition of these oils is similar to that previously published. Monoterpenes are the most important products (>95% of the total). β-Pinene and δ-3-carene are the main components (38% and 12%, respectively). Oxygenated compounds such as bornyl acetate (9%), α- and β-terpineol (1.5%) and thymol (0.45%) were also observed in smaller amounts. From the individual composition of these oils, it is concluded that all the members may be divided into two different groups (chemotypes). The first group is rich in δ-3-carene (18.7%, σ = 5.3), while the second group contains only a small percentage of this monoterpene (0.72%, σ = 0.50). The same kind of division was observed with thymol. The majority of the individual trees have a low content of this oxygenated terpene (<0.06%). Five trees have a thymol concentration between 1.5% and 3.0%. These five trees belong to groups of A. balsamea that are both rich and poor in δ-3-carene.“
[Essential oil analysis of balsam fir Abies balsamea (L.) Mill., Régimbal, J.M., Collin, G., Journal of Essential Oil Research, Vol.6(3), 1994, 229-238]
„The essential oil of A. balsamea is essentially constituted of monoterpenes (>96%) and some sesquiterpenes. β-pinene (29.9%), δ-3-carene (19.6%) and α-pinene (14.6%) were the major components.“
[Composition and antibacterial activity of Abies balsamea essential oil., Pichette, A., Larouche, P.L., Lebrun, M., Legault, J., Phytotherapy Research, Vol.20(5), 2006, 371-373]
The steam-distilled essential oil of the foliage contained β-pinene (33.5%), as main component, together with α-pinene (11.3%), δ-3-carene (14.0%), and bornyl acetate (10.0%). Minor components were camphene (5.7%), limonene (7.1%), and β-phellandrene (7.2%). Main components found in the hydrosol were α-terpineol, maltol, and bornyl acetate.
[Garneau, François-Xavier, et al. „Chemical Composition of the Hydrosol and the Essential Oil of Three Different Species of the Pinaceae Family: Picea glauca (Moench) Voss., Picea mariana (Mill.) BSP, and Abies balsamea (L.) Mill.“ Journal of Essential Oil Bearing Plants 15.2 (2012): 227-236]
Main volatiles found in the headspace of balsam fir needles powder were α-pinene, camphene, β-pinene, δ-3-carene, β-phellandrene, and bornyl acetate. Minor ones were tricyclene, β-myrcene, terpinolene, maltol, camphor, borneol, myrtenal, piperitone, longifolene, α-longipinene, β-caryophyllene, α-humulene, and α-bisabolene.
[Volatile compounds in the foliage of balsam fir analyzed by static headspace gas chromatography (HS-GC): An example of the spruce budworm defoliation effect in the boreal forest of Quebec, Canada., Caron, L., Deslauriers, A., Mshvildadze, V., Pichette, A., Microchemical Journal, 110, 2013, 587-590]
„Gas chromatography/mass spectrometry (GC/MS) of Abies balsamea, Picea mariana and Tsuga canadensis leaf essential oils assigned bornyl acetate as a major peak in A.balsamea and P.mariana, while isobornyl acetate was identified as the major peak in T.canadensis. Though these two isomers elute closely on GC, their characteristic mass spectra allow unequivocal structural assignment…
[Bernart, Matthew W. „Closely eluting bornyl and isobornyl acetates are chemotaxonomic markers in the Pinaceae by virtue of their unique mass spectra.“ American Journal of Essential Oils and Natural Products 4.2 (2016): 41-46] PDF
„The essential oil of balsam fir… [needles] is rich in monoterpenes, more than 95%. The dominant monoterpenes… are β-pinene (B), typically around 25-35%, and Δ3-carene (C) at 8-20% (with exceptions, see below). Some oxygenated compounds of this oil are bornyl acetate (F) typically between 5% and 9%, α-terpineol (D) between 0,1% and 1%, piperitone (E) between ∽0,1% and 1,2% and thymol (G) between traces and 0,3%…“ phytochemia.com

Abies balsamea (L.) Miller as Pinus balsamea L.
Woodville, W., Hooker, W.J., Spratt, G., Medical Botany, 3th edition, vol.5: t.1 (1832)
plantgenera.org

Abies balsamea cones on Niapiskau island, Mingan Archipelago National Park Reserve, Quebec, Canada © Cephas 2012 CC BY-SA 3.0 wikimedia