Wisteria floribunda (Willd.) DC. - syn. Glycine floribunda Willd.; Kraunhia floribunda (Willd.) Taub. - Fabaceae
Japanese wisteria, フジ fuji (jap.), Japanischer Blauregen, Japanische Wisteria
Deciduous woody climber, native to Japan and Korea, cultivated elsewhere as ornamental; stems twining clockwise; leaves imparipinnate, leaflets oblong; flowers in long racemes, 1.5-2cm, fragrant, corolla blue-white.
„…it has become one of the most highly romanticized flowering garden plants. It is also a common subject for bonsai, along with Wisteria sinensis…The flowers carry a distinctive fragrance similar to that of grapes.“ wikipedia
An absolute obtained from W.floribunda petals (Iwate, Japan; hexane and ethanol, 1.4g from 14kg petals) was fractionated and analyzed by GC, GC/MS, IR and NMR. The absolute had an odor similar to that of the natural wisteria flower. Main components were palmitic acid (28.5%), methyl palmitate 14.0%), 2-phenylethanol (4.4%), benzyl benzoate (3.9%), 1-phenyl-2,3-butandiol (threo 3.6%, erythro 0.8%), cinnamyl benzoate (3.3%), anisaldehyde (1.8%), and 2-phenylethyl benzoate (1.6%). „In contrast, benzyl cyanide, benzothiazole, methyl jasmonate, cis-jasmone and 3-hydroxy-4-phenyl-2-butanone were identified as the characteristic odor constituents of the flower.“
[I. Watanabe, T. Yanai, K. Awano, K. Kogami, and K. Hayashi, „Volatile components of Wisteria flower“, in „Flavors and Fragrances. A World Perspective,“ ed. by B. M. Lawrence, B. D. Mookherjee, and B. J. Wilis, Elsevier, Amsterdam, 1988, 425-437]
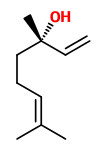
3-Hydroxy-4-phenyl-2-butanone (creamy sweet fruity) was shown to contribute strongly to the characteristic odor of wisteria flowers. „The natural ketol in wisteria flowers was characterized as a 5:1 mixture of (S)-form and (R)-form.“
[Awano, Ken-Ichi, et al. „Synthesis of all four possible stereoisomers of 1-phenyl-2,3-butanediol and both enantiomers of 3-hydroxy-4-phenyl-2-butanone to determine the absolute configuration of the natural constituents.“ Bioscience, biotechnology, and biochemistry 59.7 (1995): 1251-1254] PDF
The headspace of Wisteria floribunda flowers contained (S)-(+)-linalool (18.9%; 99.0%ee; coriandrol) with its flowery green note also known from green cardamom and jasmine absolute.
[Analysis of enantiomeric ratios of aroma components in several flowers using a Chiramix column., Tamogami, S., Awano, K.I., Amaike, M., Takagi, Y., Kitahara, T., Flavour and fragrance journal, 19(1), 1-5, 2004]
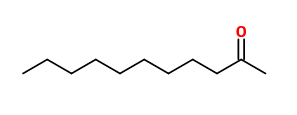
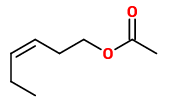
Of the 22 volatiles emitted from Japanese wisteria flowers and collected using a dynamic headspace technique, linalool (11.2µg/h/g), (E)-β-ocimene (4.0), benzyl alcohol (2.7) and 2-undecanone (0.9) were the most abundant. Sesquiterpenes including α-copaene, (E)-β-caryophyllene, α-humulene, germacrene D, and elemol were exclusively detected from japanese wisteria, whereas (E)-β-farnesene was detected from both chinese wisteria and japanese wisteria. Other compounds present in W.floribunda flower scent were (Z)-3-hexenyl acetate, methyl hexadecanoate, nonanal, decanal, indole, methyl chavicol, benzyl isocyanide and 1-nitro-2-phenylethane.
[Floral scent in Wisteria: chemical composition, emission pattern, and regulation., Jiang, Y., Chen, X., Lin, H., Wang, F., Chen, F., Journal of the American Society for Horticultural Science, Vol.136(5), 2011, 307-314]
PDF
The main components of the volatiles from W.floribunda flowers were ocimene, (Z)-3-hexenyl acetate, limonene, α-pinene, camphene, (E)-3-caren-2-ol, 2-nonanone, 1,8-cineole, myrcene, and benzaldehyde.
[Changes in constituents and contents of volatile organic compounds in Wisteria floribunda at three flowering stages., Wang Q., Wang D., Zhang R., Gao Y., Journal of Zhejiang A&F University, Vol.31(4), 2014, 647-653]
PDF

Wisteria floribunda, CC BY-SA 3.0, Author: Andreas Kraska