Psidium guajava L. - syn.Psidium pomiferum D.C., Psidium pyriferum L. - Myrtaceae - common guava, Echte Guave
Evergreen shrub or small tree native to the Caribbean, Central America and South America, widely cultivated for the fruit in tropical and subtropical regions around the world. „Various cultivars have white, pink, or red flesh, and a few also feature red (instead of green) skin.“
http://en.wikipedia.org/wiki/Psidium_guajava
„A study of the key sulfur odor components of guava was undertaken by headspace and solvent extraction techniques. GC and GC/MS analysis identified five sulfur components; dimethyl disulfide, dimethyl trisulfide, benzothiazole, 3-mercaptohexanol and 3-mercaptohexyl acetate. Two of these compounds have not previously been reported as occurring in guava (3-mercaptohexanol and 3-mercaptohexyl acetate). These compounds have a cassis-like odor and are thought to make an important contribution to the odor of guava.“
[Clery, Robin A., and Christopher J. Hammond. „New sulfur components of pink guava fruit (Psidium guajava L.).“ Journal of Essential Oil Research 20.4 (2008): 315-317]
The most important (FD factors) aroma compounds present in fresh, pink-fleshed Colombian guavas (Psidium guajava, L.), variety regional rojo, were 4-methoxy-2,5-dimethyl-3(2H)-furanone, 4-hydroxy-2,5-dimethyl-3(2H)-furanone, 3-sulfanylhexyl acetate, and 3-sulfanyl-1-hexanol followed by 3-hydroxy-4,5-dimethyl-2(5H)-furanone, (Z)-3-hexenal, trans-4,5-epoxy-(E)-2-decenal, cinnamyl alcohol, ethyl butanoate, hexanal, methional, and cinnamyl acetate. Enantioselective GC revealed an enantiomeric distribution close to the racemate in 3-sulfanylhexyl acetate as well as in 3-sulfanyl-1-hexanol.
[Characterization of the aroma-active compounds in pink guava (Psidium guajava, L.) by application of the aroma extract dilution analysis., Steinhaus, M., Sinuco, D., Polster, J., Osorio, C., Schieberle, P., Journal of agricultural and food chemistry, 56(11), 2008, 4120-4127]
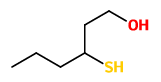 3-sulfanyl-1-hexanol (grapefruit-like) | 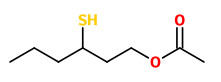 3-sulfanylhexyl acetate (tropical fruity, black currant-like) | 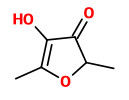 4-hydroxy-2,5-dimethyl-3(2H)-furanone (DHF, HDMF, furaneol) (sweet, cotton candy-like) |
„Seventeen aroma-active volatiles, previously identified with high flavor dilution factors in fresh, pink Colombian guavas (Psidium guajava L.), were quantified by stable isotope dilution assays. On the basis of the quantitative data and odor thresholds in water, odor activity values (OAV; ratio of concentration to odor threshold) were calculated. High OAVs were determined for the green, grassy smelling (Z)-3-hexenal and the grapefruit-like smelling 3-sulfanyl-1-hexanol [2µg/kg fruit; OAV 9300] followed by 3-sulfanylhexyl acetate (black currant-like), hexanal (green, grassy), ethyl butanoate (fruity), acetaldehyde (fresh, pungent), trans-4,5-epoxy-(E)-2-decenal (metallic), 4-hydroxy-2,5-dimethyl-3(2H)-furanone (caramel, sweet), cinnamyl alcohol (floral), methyl (2S,3S)-2-hydroxy-3-methylpentanoate (fruity), cinnamyl acetate (floral), methional (cooked potato-like), and 3-hydroxy-4,5-dimethyl-2(5H)-furanone (seasoning-like). Studies on the time course of odorant formation in guava puree or cubes, respectively, showed that (Z)-3-hexenal was hardly present in the intact fruits, but was formed very quickly during crushing. The aroma of fresh guava fruit cubes, which showed a very balanced aroma profile, was successfully mimicked in a reconstitute consisting of 13 odorants in their naturally occurring concentrations. Omission tests, in which single odorants were omitted from the entire aroma reconstitute, revealed (Z)-3-hexenal, 3-sulfanyl-1-hexanol, 4-hydroxy-2,5-dimethyl-3(2H)-furanone, 3-sulfanylhexyl acetate, hexanal, ethyl butanoate, cinnamyl acetate, and methional as the key aroma compounds of pink guavas.“
[Characterization of the key aroma compounds in pink guava (Psidium guajava L.) by means of aroma re-engineering experiments and omission tests. Steinhaus, M., Sinuco, D., Polster, J., Osorio, C., Schieberle, P., Journal of agricultural and food chemistry, Vol.57(7), 2009, 2882-2888]
„For pink- as well as for white-fleshed guavas, it was found that unripe fruits were characterised by predominant grassy and fresh notes. During ripening a fruity odour note was developed, but at the overripe stage the characteristic sulphury odour note was less intense.
…numerous compounds are formed during guava ripening process, but only few of them are responsible for the characteristic fruit flavour… (Z)-3-hexenal, ethyl butanoate, 4-hydroxy-2,5-dimethyl-3(2H)-furanone, 3-sulphanyl-1-hexanol, and 3-sulphanylhexyl acetate were established as major odour-active compounds in guava at al ripening stages, but nevertheless their concentrations differed significantly depending on maturity… 3-sulphanylhexyl acetate and 3-sulphanyl-1-hexanol decreased with ripening, while the concentrations of aliphatic esters and furanones increased during this process“
[Changes in odour-active compounds of two varieties of Colombian guava (Psidium guajava L.) during ripening., Sinuco, D.C., Steinhaus, M., Schieberle, P., Osorio, C., European Food Research and Technology, 230(6), 2010, 859-864]
Guava powders obtained by hot-air drying (G-HA) or lyophilisation (G-LF) processes lost the key aroma compounds of guava, such as (Z)-3-hexenal, hexanal, 3-sulfanyl-1-hexanol, 3-sulfanylhexyl acetate, 4-hydroxy-2,5-dimethyl-3(2H)-furanone, and ethyl butanoate, during drying processes.
[Thermal and structural study of guava (Psidium guajava L) powders obtained by two dehydration methods., Osorio, C., Carriazo, J.G., Barbosa, H., Química Nova, 34(4), 2011, 636-640] http://www.scielo.br/scielo.php?pid=S0100-40422011000400016&script=sci_arttext&tlng=pt
Of the volatile compounds of guava wine isolated by continuous solvent extraction and analyzed by GC-FID and GC-MS, twelve were considered as odor-active: (E)-β-damascenone, ethyl octanoate, ethyl 3-phenylpropanoate, ethyl hexanoate, 3-methylbutyl acetate, 2-methyltetrahydrothiophen-3-one, 2,5-dimethyl-4-methoxy-3(2H)-furanone, ethyl (E)-cinnamate, ethyl butanoate, (E)-cinnamyl acetate, 3-phenylpropyl acetate, and ethyl 2-methylpropanoate.
[Pino, Jorge A., and Oscar Queris. „Characterization of odor-active compounds in guava wine.“ Journal of agricultural and food chemistry 59.9 (2011): 4885-4890]
(E)-β-ionone, ethyl hexanoate, ethyl butanoate, hexanal, (Z)-3-hexenal, hexyl acetate, (E)-2-hexenal and limonene were found to contribute most to the typical guava aroma of Psidium guajava L. cv. Red Suprema.
[Odour‐active compounds in guava (Psidium guajava L. cv. Red Suprema)., Pino, J. A., Bent, L., Journal of the Science of Food and Agriculture, 93(12), 2013, 3114-3120]
Psidium guajava as Psidium pumilum Vahl; Blanco, M., Flora de Filipinas, t.172 (1875)
http://plantgenera.org/species.php?id_species=844853

Guava fruits, Kadavoor, Kerala, India https://commons.wikimedia.org/wiki/File:Psidium_guajava_at_Kadavoor.jpg
by Jee & Rani Nature Photography (2015) CC BY-SA 4.0
