Phlox paniculata L. - Polemoniaceae - garden phlox, Hoher Stauden-Phlox, Rispige Flammenblume, Herbstflieder
Perennial herb, 50-120cm high, native to eastern North America, along rivers and open woods, extensively cultivated as ornamental.
http://plants.usda.gov/core/profile?symbol=PHPA9
„The flowers, 1.5-2.5cm in diameter, often fragrant, are white, pink, red, or purple, and borne in summer through fall (autumn). The flowers are grouped in panicles, hence the specific epithet paniculata.“
http://en.wikipedia.org/wiki/Phlox_paniculata
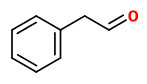
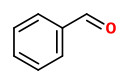
Main compounds of a dynamic headspace concentrate from violet Phlox flowers were phenylacetaldehyde (20%), benzaldehyde (5%), lilac alcohols (3%), linalool (2%), and 2-phenylethanol (0.4%).
[Volatile constituents of European bird cherry flowers (Padus avium Mill.). Surburg, H., Güntert, M., Schwarze, B.,Journal of Essential Oil Research, Vol.2(6), 1990, 307-316]
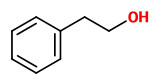
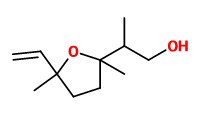
The fragrant volatiles of Phlox paniculata hybrids were collected by dynamic and vacuum headspace analysis. The product of the vacuum process contained mainly 2-phenyl ethanol (12-17%) and linalool (3-9%), together with 3-hydroxy-1-phenyl-2-butanone (1-4%), 3-hydroxy-4-phenyl-2-butanone (3-7%) and 1-phenyl-2,3-butanediol (0.3-6%). Cinnamic alcohol was present only in the vaccum product. Phenylacetaldehyde (1-2%), benzaldehyde (0.3-5%) and benzyl alcohol (0.2-2%) have been found both in the dynamic headspace and in the vaccum headspace product. Furthermore, lilac alcohols, eugenol methyl ether, isoeugenol methyl ether and germacrene-D are additional volatiles of Phlox flowers.
[Surburg et al., Volatile compounds from Flowers, in: Teranishi, R.;Buttery, R. G.;Sugisawa, H. Bioactive volatile compounds from plants. (Book) 1993, 170-171]
The flower scent collected using the dynamic headspace adsorption method and analysed by GC-MS showed benzaldehyde (1.5%), phenylacetaldehyde (17.3%), 2-phenylethanol (10.2%), eugenol (1.5%), (Z)-β-ocimene (1.6%), (E)-β-ocimene (40.8%), linalool (3.8%), lilac alcohol A (1.2%), β-cubebene (1.6%), germacrene D (5.5%), 3-methylbutylnitrile (1.0%), 3-methyl syn-butyl aldoxime (2.0%), and 3-methyl anti-butyl aldoxime (1.1%) as main components. Minor components were e.g. methyl benzoate (0.1%), methyl salicylate (0.3%), benzyl alcohol (0.2%), myrcene (0.9%), the lilac aldehydes A/B/C/D (0.4/0.3/0.2/0.1%), lavandulol (0.1%), and the lilac alcohols B/C/D (0.1/0.2/0.6%).
[Floral scents in butterfly‐pollinated plants: possible convergence in chemical composition., Andersson, S., Nilsson, L.A., Groth, I., Bergström, G., Botanical Journal of the Linnean Society, 140(2), 2002, 129-153]
Scent emission analysis of P.paniculata flowers showed the benzenoids, benzaldehyde (14-91 ng/hr/flower), 2-phenylacetaldehyde (14-61 ng/hr/flower) and methyl eugenol (11-47 ng/hr/flower) as main constituents, together with (E)-β-ocimene (13-28 ng/hr/flower). Other components were linalool, lilac aldehyde A, lilac aldehyde B, 2-phenyl acetonitrile, eugenol and methyl isoeugenol as well as nonanal and decanal.
[Floral odor bouquet loses its ant repellent properties after inhibition of terpene biosynthesis., Junker, R.R., Gershenzon, J., Unsicker, S.B., Journal of chemical ecology, 37(12), 2011, 1323-1331]

Step,E., Bois,D., Favourite flowers of garden and greenhouse, vol.3 t 185 (1896-1897) [D.G.J.M. Bois]
http://plantgenera.org/species.php?id_species=780349

garden phlox, CC BY-SA 3.0, Author: Andreas Kraska