Philadelphus coronarius L. - Hydrangeaceae
(sweet) mock-orange, English dogwood, Seringat, Jasmin des poètes (fr.), Europäischer Pfeifenstrauch, Falscher Jasmin
Deciduous shrub, up to 4m tall, native from Central and Southeast Europe to Western Asia, cultivated and naturalized elsewhere; leaves opposite, oval, dentate; racemes of white to yellowish white flowers, 2.5-3.5cm in diam., strongly aromatic.
„It is a popular ornamental plant for gardens in temperate regions, valued for its profuse sweetly scented white blossom in early summer.“ http://en.wikipedia.org/wiki/Philadelphus_coronarius
The pre-female 'nectar producing disk flowers' are pleasantly fragrant, especially in the evening and then particularly intense. http://de.wikipedia.org/wiki/Philadelphus_coronarius
Major constituents of the vacuum headspace concentrate of the flower volatiles were acetophenone (20%), linalool oxides (11.5%), and indole (10%). Among the minor constituents were 2-aminobenzaldehyde (2%), benzothiazole (1%), jasmone (trace), and (Z)-hexenyl benzoate (trace).
[Joulain, D. „Study of the fragrance given off by certain springtime flowers.“ Progress in essential oil research (1986): 57-67]
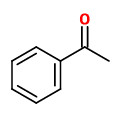 acetophenone | 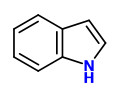 indole (sweet floral) | 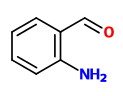 2-aminobenzaldehyde (sweet orange flower) | 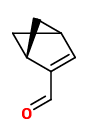 myrtenal (sweet spicy) | 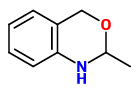 1,4-dihydro-2-methyl-2H-3,1-benzoxazine (quinoline-like) |
Trans-myrtanal (46%), cis-myrtanal (2.2%), myrtenal, myrtenol, indole, nopinone (5.3%), 2-aminobenzaldehyde (11%), 2-formamidobenzaldehyde and veratraldehyde have been determined in the headspace of living flowers by GC-MS analysis. Flowers from another single plant showed an entirely different composition with hugh amounts of acetophenone (20%) and indole (10%).
[The composition of the headspace from fragrant flowers: Further results., Joulain D., Flavour and Fragrance J 1987; 2: 149-155]
1,4-Dihydro-2-methyl-2H-3,1-benzoxazine (odor: quinoline-like) as the cyclic product of anthranilic alcohol and acetaldehyde is found in the vacuum headspace concentrate of a P.coronarius flowers variety in amounts of 0.2-1%. In pure form it cleaves acetaldehyde in perceptible amounts.
[Volatile constituents of European bird cherry flowers (Padus avium Mill.). Surburg, H., Güntert, M., Schwarze, B.,Journal of Essential Oil Research, Vol.2(6), 1990, 307-316]
Feeding experiments with 13C- and fluorine-labelled precursors revealed that the biosynthesis of 2-aminobenzaldehyde in flowers of Robinia pseudoacacia and Philadelphus coronarius is realized by transformation of anthranilic acid to indole followed by oxidative ring opening and hydrolysis of the resulting N-formyl-2-aminobenzaldehyde.
[Biosynthesis of 2-aminobenzaldehyde in flowers of Robinia pseudoacacia and Philadelphus coronarius., Spiteller, P., Steglich, W., Phytochemistry, Vol.57(3), 2001, 361-363]
„The volatile constituents of both dried and fresh leaves, twigs and flowers of Philadelphus coronarius L. (Hydrangeaceae) were isolated by hydrodistillation and analyzed by GC/FID and GC/MS. There were major differences between the various oils. For example, epi-13-manool (47.6 %) was the major component of the fresh flower oil, but was not detected in the dried fower oil, whereas isolongifolol (15.5%), 2-nonanol (11.1%) and 7-hydroxycoumarin (9.7%) were the principal components of the dried flower oil. In the oils of both the fresh and dried leaves the major constituent was (E,E)-farnesol, although the proportion of this compound in the fresh leaf oil was considerably higher (37.3%) than that of the dried leaf oil (3.2%). (E,E)-farnesol was also the major component of the fresh twig oil (16.6%), but was absent from the dried twig oil, the major constituent of which was (E,E)-2,4-decadienal (2.4%); however, this was not detected in the fresh twig oil.“
[Identification of the components of Philadelphus coronarius L. essential oil., Czigle, Szilvia, et al., Journal of Essential Oil Research, Vol.18(4), 2006, 423-426]

Botanical Magazine, Vol.11, t.391 (1797)
http://plantgenera.org/species.php?id_species=777547

mock orange flowers, CC BY-SA 3.0, Author: Andreas Kraska
