Fumaria officinalis L. - Papaveraceae (also placed in Fumariaceae) - common fumitory, Gemeiner Erdrauch
Annual herb, up to 80cm tall, native to Europe, Southwest Asia, naturalized elsewhere (Northern America); leaves blueish green, deeply dissected; flowers characteristic, purplish pink or white near base, deep reddish purple to maroon apically. http://efloras.org/florataxon.aspx?flora_id=1&taxon_id=220005380
Total alkaloid content in F.offinalis aerial parts was found 1386 mg/100g d.w. (138.6?) with main components fumarophycine (19-37 mg/100g dw), O-methyl fumarophycine (17-29 mg/100g dw), and protopine (17-22 mg/100g dw). Total phenolics content in F.officinalis was 2600-2900 mg/100g dw. (?) Fumaric acid was found in all of 8 Fumaria species.
[Alkaloids and organic acids content of eight Fumaria species., Soušek, J., Guedon, D., Adam, T., Bochořáková, H., Taborska, E., Valka, I., Šimánek, V., Phytochemical Analysis, 10(1), 1999, 6-11]
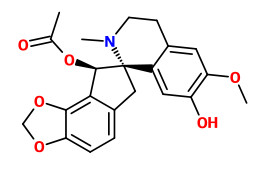
fumarophycine
(Structure of Fumarophycine., Castillo, M., Saunders, J.K., MacLean, D.B., Mollov, N.M., Yakimov, G.I., Canadian Journal of Chemistry, 49(1), 1971, 139-142)
Protopine exhibited the known papaverine-like musculotropic (antispasmodic) action. Electric field stimulated contractions of isolated ileum of guinea-pig were antagonized by protopine more than by coptisine and chelidonine.
[Antispasmodic and relaxant activity of chelidonine, protopine, coptisine, and Chelidonium majus extracts on isolated guinea-pig ileum., Hiller, K.O., Ghorbani, M., Schilcher, H., Planta medica, 64(8), 1998, 758-760]
The herb is traditionally used as cholagogue and spasmolytic to treat biliary and dyspeptic disorders. It is also used as diuretic and laxative, and externally to treat psoriasis and chronic eczema. „In common with other members of the poppy family, fumitory herb contains several alkaloids (1%): protoberberine (scoulerine) and protopine (= fumarin, the main alkohol), also spirobenzylisoquinoline (fumaricine, fumariline) and indenbenzazepines (fumaritine, fumarofine) alkaloids… The alkaloids have sedative and spasmolytic properties.“
[Medicinal Plants of the World. Ben-Erik Van Wyk and Michael Wink, Pretoria 2004, 148]
Uusing ethanol 50% (w/w) at a temperature of 79 °C with a 3% solid-liquid (w/w) ratio, the weight of protopine extracted from 100 g of F. officinalis was about 230 mg.
[Solid-liquid extraction of protopine from Fumaria officinalis L. - Experimental study and process optimization., Rakotondramasy-Rabesiaka, L., Havet, J.L., Porte, C., Fauduet, H., Separation and Purification Technology, 59(3), 2008, 253-261]
„In the 1950s fumaric acid esters were successfully used for the first time by the German chemist Schweckendiek in the self-treatment of psoriasis vulgaris. The attempt was made based on the erroneous notion that a disruption of the citrate acid cycle, whose intermediate product is fumaric acid, is essential to the pathogenesis of psoriasis. It was not until 1994 that a mixture of fumaric acid esters was approved under the name Fumaderm® by the German FDA. Nowadays the preparation is a standard treatment in German-speaking regions for control of moderate to severe psoriasis vulgaris. Seventeen years after its approval, several molecular mechanisms have been discovered underlying DMF's effect as a pharmacologically effective ester of fumaric acid.“
[Dimethyl fumarate–only an anti‐psoriatic medication?., Meissner, M., Valesky, E.M., Kippenberger, S., Kaufmann, R., JDDG: Journal der Deutschen Dermatologischen Gesellschaft, 10(11), 2012, 793-801] http://onlinelibrary.wiley.com/doi/10.1111/j.1610-0387.2012.07996.x/full

Dietrich, A.G., Flora regni borussici, vol.5 t.348 (1837)
http://plantgenera.org/species.php?id_species=446862

