Evernia prunastri (L.) Ach. - Parmeliaceae - oakmoss, Eichenmoos, Pflaumenflechte
„… a species of lichen … can be found in many mountainous temperate forests throughout the Northern Hemisphere, including parts of France, Portugal, Spain, North America, and much of Central Europe. Oakmoss grows primarily on the trunk and branches of oak trees, but is also commonly found on the bark of other deciduous trees and conifers such as fir and pine… its fragrant compounds are extracted as oakmoss absolutes and extracts. These raw materials are often used as perfume fixatives and form the base notes of many fragrances.“ http://en.wikipedia.org/wiki/Evernia_prunastri
„Oakmoss extracts possess an intense mossy-woody odor of some humid tonality, with distinct phenolic, smoky, and animalic side-notes… Orcinol 3, the mono-methyl ether of orcin, which has a woody, mossy, phenolic scent, constitutes the main part of the oakmoss complex… Most important for the typical odor character of oakmoss extracts are, however, the 2-methyl derivatives of orcin, namely 3-methoxy-2,5-dimethylphenol and Evernyl (methyl 2,4-dihydroxy-3,6-dimethylbenzoate)…“
[Scent and Chemistry, Günther Ohloff, Wilhelm Pickenhagen, Philip Kraft, Wiley-VCH, 2012, 317-321]
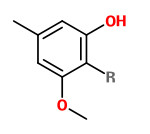 3-methoxy-5-methylphenol (Orcinyl 3, R=H) 3-methoxy-2,5-dimethylphenol (R=Me) | 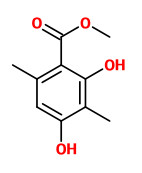 Evernyl (methyl-β-orcinol carboxylate, veramoss, verymoss) |
„…oak moss absolute, prepared from the lichen Evernia prunastri (L.) Arch., is considered a major contact sensitizer and is therefore included in the fragrance mix used for diagnosing perfume allergy… results showed that atranol and chloroatranol, formed by transesterification and decarboxylation of the lichen depsides, atranorin and chloroatranorin, during the preparation of oak moss absolute, are strong elicitants in
most patients sensitized to oak moss. Methyl-β-orcinol carboxylate, a depside degradation product and the most important monoaryl derivative of oak moss from an olfactory standpoint, was also found to elicit a reaction in most patients.“
[Contact allergy to oak moss: search for sensitizing molecules using combined bioassay-guided chemical fractionation, GC-MS, and structure-activity relationship analysis. Guillaume Bernard,
Elena Giménez-Arnau, Suresh Chandra Rastogi, Siri Heydorn, Jeanne Duus Johansen, Torkil Menné, An Goossens, Klaus Andersen, Jean-Pierre Lepoittevin, Archives of Dermatological Research, 2003, Vol. 295 (6), 229-235]
One of the most common lichen substances is (+)-usnic acid, which occurs in high amounts in E.prunastri, but is not detected in Pseudevernia furfuracea (treemoss). It is possible to isolate >50g of pure usnic acid from 350 g of a resinoid, but the absolutes are practically free of it.
Methyl-β-orcinol carboxylate (10-20%, w/w of resinoid or absolute) is the most important compound of the depsides and mono-aromatic compounds in oakmoss extracts. „Although methyl β-orcinolcarboxylate was initially described as odourless, it became rapidly obvious that it is the major contributor to the odour of oakmoss extracts. Strangely enough, one had to wait until the middle of the 1960s to see a number of processes appear in the patent literature for the production of methyl-β-orcinol carboxylate and other structurally related artificial resorcylates with possible applications in fragrance formulations.“
[Joulain, Daniel, and Raphaël Tabacchi. „Lichen extracts as raw materials in perfumery. Part 1: oakmoss.“ Flavour and fragrance journal 24.2 (2009): 49-61] https://core.ac.uk/download/pdf/20644842.pdf
„… the composition of the volatiles from the acetone soluble fractions of the methanol extracts of the lichen species: Hypogymnia physodes, Evernia prunastri and Parmelia sulcata, growing on the same host tree, collected at the same time and locality, are reported.“ Main components of the extract from E.prunastri were methyl β-orcinolcarboxylate (atraric acid; 30.1%), orcinol (25.0%), usnic acid (11.4%), methyl 2,4-dihydroxy-6-methylbenzoate (methyl orsellinate; 10.2%), 3-methoxy-5-methylphenol (orcinol monomethyl ether; 5.7%) and 2,6-dihydroxy-4-methylbenzaldehyde (atranol; 2.1%).
[Stojanović, Igor Ž., et al. „Volatile constituents of selected Parmeliaceae lichens.“ Journal of the Serbian Chemical Society 76.7 (2011): 987-994] http://www.doiserbia.nb.rs/img/doi/0352-5139/2011/0352-51391100087S.pdf

Evernia prunastri on Prunus cerasifera
CC BY-SA 3.0, Author: la la means I love you Wikimedia Commons
