Daucus carota L. subsp.sativus - Apiaceae - carrot , Möhre, Karotte, Mohrrübe
Erect biennial herb, 30-100cm high, cultivated, taproot used as vegetable. http://en.wikipedia.org/wiki/Carrot
The woody taproot of the Central European wild carrot, Daucus carota L. subsp.carota, is white and carotin-free. Today's root vegetable, Daucus carota subsp. sativus is probably result of crossing with the Southern European ssp. maximus and the oriental ssp. afghanicus, and cultivar selection.
http://www.biozac.de/biozac/capvil/Cvdaucus.htm
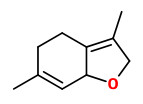
GC-trapping experiments with the aroma oil obtained by solvent extraction-steam distillation of carrots showed 2-sec-butyl-3-methoxypyrazine to be present above its detection threshold. „It exerts its effect in carrot by imparting the slightly sharp, raw, earthy, rooty character to the aroma, which complements the sweeter, oily, perfumyey contributions of the major terpenoid constituents.“
[Cronin, Denis A., and Philip Stanton. „2‐Methoxy‐3‐sec‐butylpyrazine - an important contributor to carrot aroma.“ Journal of the Science of Food and Agriculture 27.2 (1976): 145-151]
By headspace examination of the salt-saturated juice from raw vegetable tissues, 2-sec-butyl-3-methoxypyrazine has been found an important olfactory (green earthy vegetable-like) volatile component of carrot root and many other fresh vegetables like beans, beetroot, chard, lettuce, nasturtium, parsnip, pea shells, bell peppers and chili.
[Murray, Keith E., and Frank B. Whitfield. „The occurrence of 3‐alkyl‐2‐methoxypyrazines in raw vegetables.“ Journal of the Science of Food and Agriculture 26.7 (1975): 973-986]
Main volatile components of fresh carrot leaves essential oil (hydrodistilled, 0.07% yield) were sabinene (10.9%), linalool (14.9%), linalyl acetate (8.3%), carvone (8.7%) and carotol (9.8%).
[Khanna, R. K., O. S. Sharma, and Singh Akhileshwar. „The essential oil from the leaves of Daucus carota Linn. var. sativa.“ Proceedings of the 11th international congress of essential oils, fragrances and flavours. New Delhi, India, 12-16 November, 1989 Vol. 4 Chemistry-analysis and structure.. Aspect Publishing, 1990]
„Major volatile compounds identified in raw carrots were α-pinene, sabinene, myrcene, limonene, γ-terpinene, terpinolene, β-caryophyllene and γ-bisabolene. Mono-and sesquiterpenes accounted for about 97% of the total volatiles identified. Sizeable varietal differences (p<0.01) were observed. Carrot volatiles did not change appreciably during the 28 day storage period at 5, 25 and 35°C, except propanol that showed exponential increases at higher temperatures. No propanol was detected in fresh raw carrots. Cooking resulted in 88.6, 93.0 and 95.5% loss in total volatiles after cooking times of 10, 20 and 30 min, respectively.“
[Method for the static headspace analysis of carrot volatiles. C. Alasalvar, J.M. Grigor, P.C. Quantick, Food Chemistry, Volume 65, Issue 3, May 1999, Pages 391–397]
The three bitter tasting bisacetylenes [Z]-heptadeca-1,9-dien-4,6-diyne-3-ol (falcarinol), [Z]-heptadeca-1,9-dien-4,6-diyne-3,8-diol (falcarindiol) and [Z]-3-acetoxy-heptadeca-1,9-dien-4,6-diin-8-ol (falcarindiol-3-acetate) were identified in carrots and carrot purée. Especially falcarindiol was shown to be responsible for the perceived bitterness of carrot products.
[Objektivierung des bitteren Fehlgeschmacks von Karotten (Daucus carota L.) und Karottenprodukten, Czepa, A., (Doctoral dissertation, Universität München), 2004] https://mediatum.ub.tum.de/doc/601340/601340.pdf
http://www.dfal.de/fileadmin/aif/AIF-FV12226N.pdf
„Carrots (Daucus carota L.) of cv. Bolero and cv. Carlo were processed into shreds and stored for up to 4 months at −24 °C (frozen storage), or the roots were stored for up to 4 months at 1 °C (refrigerated storage) followed by processing into shreds. Volatiles from the carrot shreds were collected by dynamic headspace technique and analyzed by GC-FID, GC-MS, GC-MS/MS, and GC-O to determine the volatile composition and aroma active components of carrots stored under different temperature conditions. A total of 52 compounds were quantified, of which mono- and sesquiterpenes accounted for ∼99% of the total volatile mass. Major volatile compounds were (−)-α-pinene, β-myrcene, (−)-limonene, (+)-limonene, (+)-sabinene, γ-terpinene, p-cymene, terpinolene, β-caryophyllene, α-humulene, and (E)- and (Z)-γ-bisabolene. A considerable increase in the concentration of mono- and sesquiterpenes was observed during refrigerated storage, whereas the concentration of terpenoids was around the same level during frozen storage. GC-O revealed that the major volatiles together with (+)-α-pinene, (−)-β-pinene, (+)-β-pinene, 6-methyl-5-hepten-2-one, (−)-β-bisabolene, β-ionone, and myristicin had an odor sensation, which included notes of “carrot top”, “terpene-like”, “green”, “earthy”, “fruity”, “citrus-like”, “spicy”, “woody”, and “sweet”. “
[Changes in volatile compounds of carrots (Daucus carota L.) during refrigerated and frozen storage., Kjeldsen, F., Christensen, L.P., Edelenbos, M., Journal of agricultural and food chemistry, Vol.51(18), 2003, 5400-5407]
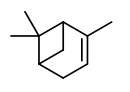
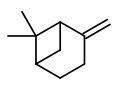
Carrot seeds yield an essential oil with antifungal activity against plant pathogenic fungi like Alternaria alternata. The main component (38.8%) and antifungal active principle is the pleasant smelling carotol. Other components are β-caryophyllene (10.7%), caryophyllene oxide (4.3%) and a second daucane sesquiterpene alcohol, daucol (2.0%).
[Antifungal activity of the carrot seed oil and its major sesquiterpene compounds., Jasicka-Misiak, I., Lipok, J., Nowakowska, E.M., Wieczorek, P.P., Młynarz, P., Kafarski, P., Zeitschrift für Naturforschung C, 59(11-12), 2004, 791-796] http://www.znaturforsch.com/ac/v59c/s59c0791.pdf
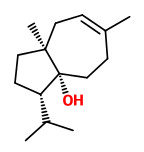
„Evaluation of the GC effluent from cooked carrot volatiles using aroma extract dilution analysis (AEDA) found linden ether with the highest flavor dilution (FD) factor. Others with 10-fold lower FD factors were β-ionone, eugenol, the previously unidentified β-damascenone, (E)-2-nonenal, octanal (+ myrcene), and heptanal. All other previously identified volatiles showed lower FD factors. Odor thresholds, concentrations, and odor activity values of previously identified compounds are reviewed. This indicated that at least 20 compounds occur in cooked carrots above their odor thresholds (in water). Compounds showing the highest odor activity values included β-damascenone, (E)-2-nonenal, (E,E)-2,4-decadienal, β-ionone, octanal, (E)-2-decenal, eugenol, and p-vinylguaiacol.“
[Cooked Carrot Volatiles. AEDA and Odor Activity Comparisons. Identification of Linden Ether as an Important Aroma Component. Ron G. Buttery and Gary R. Takeoka, J. Agric. Food Chem., 2013, 61 (38), pp 9063–9066]
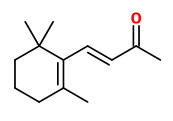
Like in melon, watermelon, tomato, and pepper, there is an association between carotenoid pigmentation and
aroma volatile content. Neither β-ionone nor α-ionone and β-cyclocitral have been found in the cultivars 'Yellowstone' (yellow) and 'Creme de Lite' (white). Orange cultivars like 'Nairobi', 'Rothild', and the purple-orange 'Purple haze' contained (μg/g f.w.) β-ionone (0.12-0.21), α-ionone (0.08-0.12), and β-cyclocitral (0.01-0.03). Farnesylacetone as a norisoprene derived from the colorless carotenoid biosynthetic intermediates phytoene or phytofluene was identified primarily in Nairobi, Rothild, and Purple Haze cultivars (2.1-2.5 μg/g f.w.).
[Formation of norisoprenoid flavor compounds in carrot (Daucus carota L.) roots: characterization of a cyclic-specific carotenoid cleavage dioxygenase 1 gene., Yahyaa, M., Bar, E., Dubey, N. K., Meir, A., Davidovich-Rikanati, R., Hirschberg, J., Lewinsohn, E., Journal of agricultural and food chemistry, 61(50), 2013, 12244-12252] http://naldc.nal.usda.gov/download/58545/PDF
The main constituents of an essential oil (0.78-0.93%) hydrodistilled from carrote seed waste treated with an enzyme preparation (derived from the nonpathogenic filamentous fungus Mucor circinelloides) were: Carotol (34.6%), sabinene (8.0%), alpha‐pinene (6.4%) and daucol (4.3%).
[Śmigielski, Krzysztof B., et al. „The Effect of Enzyme‐Assisted Maceration on Bioactivity, Quality and Yield of Essential Oil from Waste Carrot (Daucus carota) Seeds.“ Journal of food quality 37.4 (2014): 219-228] https://onlinelibrary.wiley.com/doi/full/10.1111/jfq.12092
Main mono- and sesquiterpene volatile compounds found (raw material headspace-SPME-GC-MS) in different carrot varieties (Yellowstone, Creme de Lite, Nairobi, Rothild, Purple haze) were (μg/g f.w.) β-caryophyllene (0.62-1.04), terpinolene (0.08-0.74), β-myrcene (0.16-0.68), sabinene (0.03-0.60), γ-terpinene (0.16-0.49), p-cymene (0.08-0.19), α-pinene (0.08-0.22), β-pinene (0.06-0.18), and α-humulene (0.06-0.12).
[Identification and characterization of terpene synthases potentially involved in the formation of volatile terpenes in carrot (Daucus carota L.) roots., Yahyaa, M., Tholl, D., Cormier, G., Jensen, R., Simon, P.W., Ibdah, M., Journal of agricultural and food chemistry, 63(19), 2015, 4870-4878] http://www.faculty.biol.vt.edu/tholl/publications/Yahyaa_etal_2015.pdf

Lindman,C.A.M., Bilder ur Nordens Flora, vol.2 t.254 (1922-1926)
http://plantgenera.org/species.php?id_species=322161
Daucus carota ssp. sativus
© Rolf Marschner (2005),
www.botanische-spaziergaenge.at