Cydonia oblonga Mill. - Cydonia vulgaris Pers.; Pyrus cydonia L. - Rosaceae - quince, Quitte
Deciduous shrub or small tree, to 8m high, native to West Asia, Caucasus, naturalized and cultivated elsewhere; branchlets purplish red when young, purplish brown when old; leaves ovate to oblong, densely villous; flowers white or pinkish, 4–5 cm in diam.
http://www.efloras.org/florataxon.aspx?flora_id=2&taxon_id=200010815
„Marmelade“ for jam is derived from Portuguese marmelo for quince, from the Greek melimelon „honey apple“.
http://de.wikipedia.org/wiki/Quitte
„Quince cheese is prepared with quince fruits. The fruit is cooked with sugar, turns red after a long cooking time, and becomes a relatively firm quince jelly, dense enough to hold its shape. The taste is sweet but slightly astringent, and it is similar in consistency, flavor and use to guava cheese or guava paste.“
https://en.wikipedia.org/wiki/Quince_cheese
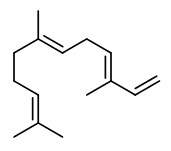
The essential oil (yield 0.004~0.017%) obtained by steam distillation of fresh C.oblonga from Japan had a strong fruity aroma and consisted mainly of trans-α-farnesene (58%). Furthermore, ethyl esters of saturated and unsaturated fatty acids, like ethyl hexanoate (4.6%), ethyl octanoate 11.2%), ethyl decanoate (4.2%) and ethyl dodecanoate were found (GC/MS).
[The constituents of the essential oil from Japanese quince fruit, Cydonia oblonga Miller., Shimizu, S., Yoshihara, S., Agricultural and Biological Chemistry, Vol.41(8), 1977, 1525-1527]
http://www.tandfonline.com/doi/pdf/10.1080/00021369.1977.10862709
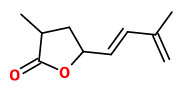
The strong sweet and fruity smelling steam-distilled (0.0058%) essential oil of Japanese quince contained the marmelo lactones and their structures have been confirmed by independant synthesis.
[Tsuneya, Tomoyuki, et al. „Isolation and identification of novel terpene lactones from quince fruit (Cydonia oblonga Mill., Marmelo).“ Agricultural and Biological Chemistry 44.4 (1980): 957-958] https://www.jstage.jst.go.jp/article/bbb1961/44/4/44_4_957/_pdf
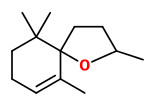
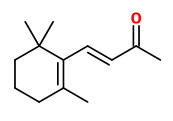
62 compounds were identified in the essential oil (steam distillation) of quince fruit, trans-α-farnesene (31.3%), (+)-trans-marmelo lactone (10.3%) and (-)-cis-marmelo lactone (13.0%) were detected as the major components. The marmelo lactones own a sweet and strong aroma and seem to contribute considerable to the main aroma of quince fruit. Some of the furfural (3.4%) probably is derived from the decomposition of precursors such as pentose and ascorbic acid in the process of steam distillation. The four stereoisomers of megastigma-4,6,8-trien-3-one (2.9%), trans-marmelo oxide (2.4%), cis-marmelo oxide (2.2%), theaspirane (2.9%), theaspirone (1.5%), vitispirane (1.0%), 3,4-dehydro-β-ionone (0.04%) and β-ionone (0.15%) contribute to quince aroma. Only small amounts (2.16%) of fruit esters are present, mostly ethyl caprylate (0.8%), ethyl caproate (0.4%) and cis-3-hexenyl caproate (0.3%).
[Volatile components of quince fruit (Cydonia oblonga Mill.)., Tsuneya, T., Ishihara, M., Shiota, H., Shiga, M., Agricultural and Biological Chemistry, Vol.47(11), 1983, 2495-2502]
http://www.tandfonline.com/doi/pdf/10.1080/00021369.1983.10865983
„Ripe quince fruits are not edible raw because of their hardness and bitterness, but they nevertheless emit a floral odor so strong that they are sometimes used as household room refreshers.“ Oils obtained from the peel of quince fruit consisted mainly (GC 77%) of esters, mostly ethyl esters.
[Volatile constituents of peel of quince fruit, Cydonia oblonga Miller., Umano, K., Shoji, A., Hagi, Y., Shibamoto, T., Journal of Agricultural and Food Chemistry, Vol.34(4), 1986, 593-596]
Theaspiranes A/B (fresh green-woody/cassis-like) are formed at natural pH of the quince fruit pulp under acidic conditions (pH 3.5-3.8) from the labile precursor 4-hydroxy-7,8-dihydro-beta-ionol.
[Winterhalter, Peter, and Peter Schreier. „4-Hydroxy-7,8-dihydro-beta-ionol: natural precursor of theaspiranes in quince fruit (Cydonia oblonga, Mill.).“ Journal of Agricultural and Food Chemistry 36.3 (1988): 560-562]
Main volatile components (HS-GC-MS) of freeze-dried fruit puree were ethanol (~50%), ethyl acetate (35.4%), ethyl hexanoate (2.8%), the theaspiranes (2.2%), ethyl octanoate (1.4%), and ethyl 2-methylbutyrate (~1%). Minor components were e.g. limonene, trimethylbenzol, alpha ionene, farnesene, acetoin, safranal, and vitispirane; esters like propyl acetate, ethyl butyrate, and ethyl (E)-2-octenoate; alcohols like isobutanol, 2-methylbutanol and hexanol; and aldehydes like hexanal, octanal and nonanal.
[Heiduk, Andrea. Einfluss verschiedener Trocknungsmethoden auf Aromen von Quitten und Scheinquitten. Diss. Hochschule, 2018, 43-47]

Thomé, O.W., Flora von Deutschland Österreich und der Schweiz, Tafeln, vol.3 t.419 (1885)
http://www.plantillustrations.org/species.php?id_species=304635
Cydonia oblonga
© Rolf Marschner (2006),
www.botanische-spaziergaenge.at