Crocus sativus L. - Iridaceae - saffron, saffron crocus, Safran, Echter Safran, Gewürzsafran
Perennial without above-ground stem, unknown as a wild plant; leaves 2-4, linear, with a white midrib, 8-15cm long, appearing in autumn with the lilac-purple flowers; anthers yellow, filaments purple; style red, branched, up to 3cm long; fruit 3-celled capsule.
„Cultivated in the main Kashmir valley. Being a triploid it is sterile and propagated by vegetative means only. Known only under cultivation; Saffron, obtained from long red style branches and stigmas are in great demand for eastern culinary preparations and in medicine. It is rich in vitamin B2.“ efloras.org The carotenoid crocin is primarily responsible for the color of saffron.
The freshly picked stigmas are nearly odorless. The typical saffron flavor is developed during the drying process. The aroma description of β-isophorone (3,5,5-trimethyl-3-cyclohexen-1-one) is „saffron, floral and hay“.
[Cadwallader, K. R. (2002). Flavor chemistry of saffron. In ACS Symposium series (Vol. 802, pp. 220-240). Washington, DC; American Chemical Society; 1999]
4-Hydroxy-2,6,6-trimethyl-1-cyclohexene-1-carboxaldehyde is a precursor of safranal and may be obtained by mild extraction methods like ultrasound-assisted extraction in amounts of 0.4mg/g saffron (safranal: 6mg/g).
[Qualitative determination of volatile compounds and quantitative evaluation of safranal and 4-hydroxy-2,6,6-trimethyl-1-cyclohexene-1-carboxaldehyde (HTCC) in Greek saffron. Kanakis, C.D., Daferera, D.J., Tarantilis, P.A., & Polissiou, M.G., Journal of agricultural and food chemistry, Vol.52(14), 2004, 4515-4521]
„Safranal, the main component of Crocus sativus essential oil, is thought to be the main cause of saffron unique odor… Beside its exclusive color, saffron presents a particular taste which is originates from its picrocrocin content. Safranal (2,6,6-trimethyl-1,3-cyclohexadien-1-carboxaldehyde) is a cyclical terpenic aldehyde produced from picrocrocin. Picrocrocin, discovered by Kajser (1884), cracks down following acids and alkali conditions, resulting in a molecule of water and an aglycon which in turn, loses a water molecule and finally turns to safranal. Kuhn and Winterstein (1933), who obtained safranal through picrocrocin hydrolyzation for the first time, named this chemical as Safranal.“
[Safranal: From an Aromatic Natural Product to a Rewarding Pharmacological Agent. Ramin Rezaee1 and Hossein Hosseinzadeh, Iran J Basic Med Sci. Jan 2013; 16(1), 2013, 12-26]
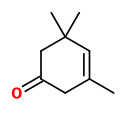 β-isophorone (saffron, floral-hay) | 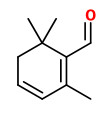 safranal (spicy phenolic) | 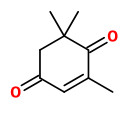 4-ketoisophorone (musty) |
From 19 saffron samples collected under different Moroccan environments, 57 volatile components have been
identified. 14 compounds were found in common to all the accessions, and seven major compounds are dominant in all samples: safranal (9-57%), 1,8-cineole (3-27%), 4-keto-isophorone (4-oxoisophorone, 3-22%), isophorone (2-20%), α-pinene (1-15%), 2,6,6-trimethyl,1,4-cyclohexadien-1-carboxaldehyde (safranal-isomer) (1-3%) and β-isophorone (0.6-6%).
Minor components present in all samples were 2(5H)-furanone, camphene, myrcene, α-phellandrene, δ-3-carene, α-terpinene, p-cymene and 4-methylene isophorone. β-Isophorone is considered one of the quality marker compounds - 70% of samples analyzed in the study contained β-isophorone.
[Phytochemical composition of Moroccan saffron accessions by headspace solid-phase-microextraction, Lage Mounira, Melai Bernardo, Cioni Pier Luigi, Flamini Guido, Gaboun Fatima, Bakhy Khadija, Zouahri Abdelmjid, Pistelli Luisa, American Journal of Essential Oils and Natural Products 2015; 2(4): 1-7]
Aroma extract dilution analysis (AEDA) was used for the determination of aroma-active compounds of Iranian saffron obtained by solvent-assisted flavour evaporation (SAFE). „According to sensory analysis, the
aromatic extract obtained by SAFE was the most representative of saffron odour…. A total of nine aroma-active compounds were detected in the aromatic extract. On the basis of the flavour dilution (FD) factor, the most powerful aroma active compounds were safranal (FD = 512), 4-ketoisophorone (FD = 256) and dihydrooxophorone (3,5,5-trimethylcyclohexane-1,4-dione; FD = 128).“
[GC-MS-olfactometric characterization of the most aroma-active components in a representative aromatic extract from Iranian saffron (Crocus sativus L.)., Amanpour, A., Sonmezdag, A.S., Kelebek, H., Selli, S., Food chemistry, 182, 2015, 251-256]
„A specification for high-quality saffron of > 20% crocins, > 6% picrocrocin and not less than 0.3% of volatiles, calculated as sum of safranal, isophorone and ketoisophorone, was developed… Saffron extracts and crocetin had a clear binding capacity at the PCP bindingside of the NMDA receptor and at the σ1 receptor, while the crocins and picrocrocin were not effective.“
[Lechtenberg, Matthias, et al. „Quality and functionality of saffron: quality control, species assortment and affinity of extract and isolated saffron compounds to NMDA and σ1 (sigma-1) receptors.“ Planta medica 74.07 (2008): 764-772]
„Saffron is the dried stigma of Crocus sativus L. and has been used for centuries in traditional medicine mainly for its healing properties… Recent phytochemistry and pharmacological experiments have indicated that crocin and safranal, the major active ingredients of saffron, exert important actions, such as antioxidant, anti-tumor, anti-diabetic, anti-inflammatory and anti-atherosclerotic. Unfortunately, the vast majority of those data derive from in vitro studies, whereas a limited number of in vivo experiments support the aforementioned effects.“
[Saffron: a natural product with potential pharmaceutical applications., Christodoulou, E., Kadoglou, N.P., Kostomitsopoulos, N., Valsami, G., Journal of Pharmacy and Pharmacology, 67(12), 2015, 1634-1649] PDF
Patients with major depressive disorder accompanied by anxious distress, who were randomly assigned to receive either saffron (30 mg/day) or citalopram (40 mg/day), showed significant improvement in scores of the Hamilton Rating Scale for Depression and Hamilton Rating Scale for Anxiety (P-value<0.001 in both groups).
„Comparison of score changes between the 2 trial arms showed no significant difference (P-value=0.984). Frequency of side effects was not significantly different between the 2 groups… The present study indicates saffron as a potential efficacious and tolerable treatment for major depressive disorder with anxious distress.“
[Ghajar, A., et al. „Crocus sativus L. versus Citalopram in the Treatment of Major Depressive Disorder with Anxious Distress: A Double-Blind, Controlled Clinical Trial.“ Pharmacopsychiatry (2016).]

Köhler,F.E., Medizinal Pflanzen, vol.2 t.164 (1890) plantgenera.org
Crocus sativus © Rolf Marschner (2010) botanische-spaziergaenge.at
