Crataegus monogyna Jacq., Crataegus laevigata (Poir.) DC.(Crataegus oxyacantha auct.), and Crataegus rhipidophylla Gand. (syn.Crataegus oxyacantha L.) - Rosaceae - hawthorn, Weißdorn
Deciduous shrub or small tree, native to the northern hemisphere; flowers with characteristic scent mixture (dead-animal/amine-like, soapy, almond-like).
Dry-extracts of hawthorn leaves with blossoms are used to treat decreasing cardic output.
„Several species of hawthorn have been used in traditional medicine, and there is considerable interest in testing hawthorn products for evidence-based medicine. The products being tested are often derived from C. monogyna, C. laevigata, or related Crataegus species, „collectively known as hawthorn“, not necessarily distinguishing between these species, which are very similar in appearance.“
http://en.wikipedia.org/wiki/Crataegus
„Ingredients are oligomeric procyanidins (OPC), glycosidic flavones as vitexin and vitexin rhamnosid and glycosidic flavonols such as rutin and hyperoside; tannins are also found at a level of 0.5 to 1%… Main indication is chronic myocardial insufficiency with resulting low blood pressure in stages I and II as defined by the New York Heart Association (NYHA).“
http://de.wikipedia.org/wiki/Crataegus
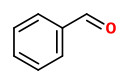
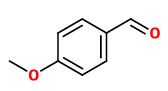
Volatiles from the flowers of hawthorn contained higher levels of benzaldehyde, 3-pyridine carboxaldehyde, anisaldehyde (4-methoxybenzaldehyde) and methyl anisate (4-methoxybenzoic acid methyl ester).
[A comparison of the flower volatiles from hawthorn and four raspberry cultivars G.W. Robertson, D.W. Griffiths, J.A.T. Woodford, A.N.E. Birch, J.A. Picketa, L.J. Wadhamsa, Phytochemistry Volume 33, Issue 5, 23 July 1993, 1047-1053]
The main fragrant volatile components in hawthorn tincture were 3-hexen-1-ol, eugenol, diethyl malate, tiglic acid and citric acid.
[Analysis of volatile fragrant components in hawthorn tincture by gas chromatography-mass spectrometry., Zhu X, Lü C, Gao Y, Tobacco Research Center, University of Science & Technology of China, Hefei 230052, China.
Wei Sheng yan jiu = Journal of Hygiene Research (2004, 33(1):108-110)].
A retrospective analysis of data from a study in which patients with mild to moderate heart failure were treated with either Crataegus Special Extract WS 1442 (CSE) 900 mg or placebo for 6 months was performed. „The primary outcome was time to progression of heart failure (HF death, hospitalisation, or sustained increase in diuretics) as assessed by log-rank tests and by Cox modelling… CSE does not reduce heart failure progression in patients who have HF. CSE appears to increase the early risk of HF progression.“
[The effect of Crataegus oxycantha special extract WS 1442 on clinical progression in patients with mild to moderate symptoms of heart failure., Zick, S.M., Gillespie, B., Aaronson, K.D., European journal of heart failure, Vol.10(6), 2008, 587-593]
http://www.ncbi.nlm.nih.gov/pmc/articles/PMC2577845/
„In this randomised, double-blind, placebo-controlled multicenter study, adults with NYHA class II or III CHF and reduced left ventricular ejection fraction (LVEF≤35%) were included and received 900 mg/day WS® 1442 or placebo for 24 months. Primary endpoint was time until first cardiac event. Results: 2681 patients (WS® 1442: 1338; placebo: 1343) were randomised. Average time to first cardiac event was 620 days for WS® 1442 and 606 days for placebo (event rates: 27.9% and 28.9%, hazard ratio (HR): 0.95, 95% CI [0.82;1.10]; p=0.476). The trend for cardiac mortality reduction with WS® 1442 (9.7% at month 24; HR: 0.89 [0.73;1.09]) was not statistically significant (p=0.269). In the subgroup with LVEF ≥ 25%, WS® 1442 reduced sudden cardiac death by 39.7% (HR 0.59 [0.37;0.94] at month 24; p=0.025). Adverse events were comparable in both groups.
Conclusions: In this study, WS® 1442 had no significant effect on the primary endpoint. WS® 1442 was safe to use in patients receiving optimal medication for heart failure. In addition, the data may indicate that WS® 1442 can potentially reduce the incidence of sudden cardiac death, at least in patients with less compromised left ventricular function.“
[The efficacy and safety of Crataegus extract WS® 1442 in patients with heart failure: The SPICE trial., Holubarsch, C.J., Colucci, W.S., Meinertz, T., Gaus, W., Tendera, M., European journal of heart failure, Vol.10(12), 2008, 1255-1263]
http://onlinelibrary.wiley.com/doi/10.1016/j.ejheart.2008.10.004/full
„Crataegus extract… WS 1442 has been demonstrated to increase thickness and reduce stiffness of the endothelial inner surface layer (the glycocalix), a sensor of blood-flow induced shear stress and modulator of endothelial NO-synthesis. The extract activates endothelial and red blood cell NO-synthase, increases endothelial NO-release and improves endothelium-dependent arterial function in animal models of aging and hypertension. The vascular effects are strongly related to the high content of oligomeric procyanidins in the extract. In addition, WS 1442 increases cardiac muscle force of contraction via an ATP-independent mechanism, has shown positive effects on remodelling in animal models of increased afterload and increases skeletal muscle mitochondrial density and fibre diameter. It has not been determined to what extent the reported vascular, cardiac and skeletal muscular effects contribute to the clinical benefit in heart failure patients… WS 1442 improved T2km (-12.7% vs. -8.4%, p = 0.019), tended to improve symptoms and to pronounce SO2-decrease with increasing exercise, an indicator of oxygen utilisation. Endurance training and WS 1442 were safe and well tolerated in combination with standard drug treatment. “
[Effects of Endurance Exercise Training and Crataegus Extract WS® 1442 in Patients with Heart Failure with Preserved Ejection Fraction–A Randomized Controlled Trial., Härtel, S., Kutzner, C., Westphal, E., Limberger, M., Burkart, M., Ebner-Priemer, U., Bös, K., Sports, Vol.2(3), 2014, 59-75]
http://www.mdpi.com/2075-4663/2/3/59/htm
Main volatile components of the essential oil obtained by hydrodistillation of the frozen flowering aerial parts (including flowers, leaves and branches) were hexanal (2.8%), (Z)-2-hexenal (1.0%), benzaldehyde (0.8%), linalool (20.3%), nonanal (2.1%), methyl salicylate (3.9%), geraniol (2.2%), β-caryophyllene (1.0%), nonadecane (13.4%), eicosane (1.9%), heneicosane (11.4%), phytol (3.0%), tricosane (5.8%), and pentacosane (1.1%). The volatile extract contained many more olfactory interesting compounds like aldehydes, theaspiranes, β-damascenone, and (Z)-jasmone. „However, the olfactive profile of the Crataegus monogyna volatile extract remains truly different from the olfactive profile of the fresh flowers.“
[Fernandez, Xavier, et al. „Forgotten perfumery plants: hawthorn volatile extract study.“ Journal of Essential Oil Research (2024): 1-12]
„In modern perfumery, the ‘hawthorn’ note is usually used in the form of p-anisaldehyde-based accords, along with coumarin, (methyl)acetophenone, anisyl alcohol, and/or p-methoxyacetophenone, to reproduce the fragrance of hawthorn's flowers. p-Anisaldehyde (4, aniseed, powdery, floral smell) was identified as a predominant compound of the volatile fraction, common to the five extracts. This observation may contribute to confirm the progressive substitution of hawthorn's natural extracts in perfumery by raw material(s) of synthetic origin, especially using synthetic anisaldehyde. Anisyl alcohol (mild-floral, aniseed, powdery smell) was also characterized in all the extracts. Acetophenone (powdery, floral notes) was only identified in the absolute of dried flowers dating from 2017. Coumarin (hay, tonka bean fragrance) was found in the two batches of absolute of dried flowers (2017 and 2018). Finally, methylacetophenone (floral smell) and p-methoxyacetophenone (floral, animal, haylike odor) were not characterized in any extracts.“
Other predominant volatile compounds were (E)-2-hexenoic acid, 2-phenylethanol, methyl salicylate, eugenol, p-methyl anisate, (Z)-jasmone, β-caryophyllene, α-farnesene, and nonadecane. Common to all extracts were also (E,E)-2,4-hexadienal, benzaldehyde, hexanoic acid, benzyl alcohol, linalool, nonanal, phenylacetonitrile, benzoic acid, (E,E,E)-2,4,6-nonatrienal, anisyl alcohol, methyl cinnamate, p-anisic acid, acetovanillone, dihydroactinidiolide, and various linear hydrocarbons (C16 to C20).
[Bouville, Anne‐Sophie, et al. „Forgotten Perfumery Plants‐Part II: New Insights into the Development of Novel Fragrant Ingredients–Hawthorn Case Study.“ Chemistry & Biodiversity 21.6 (2024): e202301020.]
https://onlinelibrary.wiley.com/doi/full/10.1002/cbdv.202301020

Klein,L., Unsere Waldbäume, Sträucher und Zwergholzgewächse, t.55 (1839) [M.Schrödter]
http://plantgenera.org/species.php?id_species=284901

Crataegus laevigata flowers, CC BY-SA 3.0, author: Andreas Kraska
Crataegus monogyna (subsp. monogyna), Wien 2017; author: Rolf Marschner,
www.botanische-spaziergaenge.at