Commiphora myrrha (Nees) Engl. - syn.Commiphora molmol Engl.; Balsamodendrum myrrha Nees - Burseraceae
(African) myrrh, Myrrhe, Somali-Myrrhenbaum
Spiny shrub or small tree, native to Northeast Africa, Arabia; fragrant resin used medicinally and as incense.
„Myrrh, a commercially used resin, is mostly from C.molmol, growing wild in Somalia, Jemen and Arabia. It is mainly used in toothpastes and tinctures for treatment of gingivitis. Myrrh is composed of the essential oil (2-10%), the ethanol soluble resin (25-40%) and the watersoluble gum (30-60%). The watersoluble gum fraction of myrrh has been found to comprise of a mixture of proteoglycans (with dominating amounts of uronic acid polymers).“ http://biomed.papers.upol.cz/pdfs/bio/2005/01/01.pdf
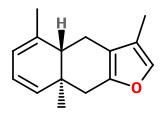
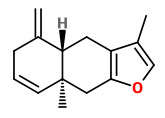
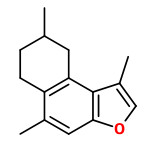
A mixture of furanoeudesma-1,3-diene and lindestrene possessed a typical myrrh odor, while dihydropyrocurzerenone possessed a resinous myrrh odor, representing the odor of myrrh by itself best.
[Wilson RA, Mookherjee BD. (1983) Characterization of aroma donating components of myrrh. Proceedings of 9th International Congress of Essential Oils, Singapore, 13-17 March, paper no.400, pp. 1-10, Book 4. Singapore: Essential Oils Association of Singapore]
From the hexan extract of myrh resin from C.molmol, 8 sesquiterpene fractions were isolated, with furanodiene, furanoeudesma-1,3-diene and curzerene, methoxyfuranodiene, acetoxyfuranodiene, curzerenone, furanodiene-6-one and methoxyfuranoguaia-9-ene-8-one, furanogermacrene-3, furanogermacrene-1 and fraction 8, furanogermacrene-2. The mixture of furanodiene-6-one and methoxyfuranoguaia-9-ene-8-one showed antibacterial and antifungal activity against standard pathogenic strains of Escherichia coli, Staphylococcus aureus, Pseudomonas aeruginosa and Candida albicans, with minimum inhibitory concentrations ranging from 0.18 to 2.8 μg/ml. These compounds also had local anaesthetic activity, blocking the inward sodium current of excitable mammalian membranes.
[Local anaesthetic, antibacterial and antifungal properties of sesquiterpenes from myrrh. Dolara, P., Corte, B., Ghelardini, C., Pugliese, A. M., Cerbai, E., Menichetti, S., Nostro, A. L., Planta medica, Vol.66(04), 2000, 356-358]
The essential oil obtained from the oleo-gum resin of C.myrrha by hydrodistillation (3.1%) was examined by using GC and GC/MS. „Among the 32 components identified in this oil, curzerene (40.1%), furanoeudesma-1,3-diene (15.0%), β-elemene (8.4%) and 2-O-acetyl-8,12-epoxygermacra-1(10),4,7,11-tetraene, isomer I (6.5%) were found to be the major constituents.“
[Constituents of the essential oil of Commiphora myrrha (Nees) Engl. var. molmol. Morteza-Semnani, K., Saeedi, M., Journal of Essential Oil Research, Vol.15(1), 2003, 50-51]
Curzerene, furanoeudesma-1,3-diene, and lindestrene were the predominant components in the headspace SPME of myrrh.
[Headspace solid phase microextraction for screening for the presence of resins in Egyptian archaeological samples. Hamm, S., Bleton, J., Tchapla, A., Journal of separation science, Vol.27(3), 2004, 235-243]
The extraction of the oleo-gum resin with supercritical carbon dioxide had a yield of 3.2%. Its main components, identified and quantified by GC/MS, were furanoeudesma-1,3-diene (34.9%), lindestrene (12.9%) curzerene (8.5%), and germacrone (5.8%). The essential oils from the same starting material by hydrodistillation (2.8%) and by steam distillation (0.4%) were similar to the SFE extract.
[Chemical composition of the essential oil and supercritical CO2 extract of Commiphora myrrha (Nees) Engl. and of Acorus calamus L. Marongiu, B., Piras, A., Porcedda, S., Scorciapino, A., Journal of agricultural and food chemistry, 53(20), 2005, 7939-7943]
„Extracts prepared from Commiphora molmol resins were analyzed by GC-MS. Twenty-two terpenoid compounds were identified in the hexane extract of the resin. Among them, 2-acetoxyfuranodiene (9.80%), furanoeudesma-1,3-diene (8.97%), isofuranogermacrene (6.71%), epicurzerenone (3.64%), 2-methoxyfuranodiene (2.97%), and lindestrene (2.74%) were the main compounds from the first myrrh resin (Tamar Ltd.), and furanoeudesma-1,3-diene (20.59%), isofuranogermacrene (17.94%), 2-acetoxyfuranodiene (8.80%), 2-methoxyfuranodiene (7.33%), and lindestrene (6.24%) from the second myrrh resin (Pamir Ltd.).“
[Fast and easy GC/MS identification of myrrh resins. Hanuš, L. O., Rosenthal, D., Řezanka, T., Dembitsky, V. M., Moussaief, A., Pharmaceutical chemistry journal, 42(12), 2008, 719-720]
„Furanoeudesma-1,3-diene possesses a sweet, warm, leathery-balsamic odor similar to that of lindestrene, while dihydropyrocurzerenone elicits a rich resinous odor impression.“
[Scent and Chemistry, Günther Ohloff, Wilhelm Pickenhagen, Philip Kraft, Wiley-VCH, 2012, 325]
To prepare pharmaceutical formulations of mouthwashes, containing extracted Yemeni myrrh as a single active constituent, different solvents were used to prepare myrrh extract. An hydroalcohol extract of myrrh with ethanol: phosphate buffer pH 7 (85:15) showed the best antimicrobial activity against Staphylococcus aureus. The selected myrrh mouthwash formulations revealed remarkable antimicrobial activity against Staph. aureus and Strep. mutans, greater than all other tested products with chlorhexidine gluconate or benzydamine HCl.
[Antimicrobial activity of Yemeni myrrh mouthwash. Almekhlafi, S., Thabit, A. A., Alwossabi, A. M., Awadth, N., Thabet, A. A., Algaadari, Z., Journal of Chemical and Pharmaceutical Research, Vol.6(5), 2014, 1006-1013] http://jocpr.com/vol6-iss5-2014/JCPR-2014-6-5-1006-1013.pdf
Main components of the Commiphora myrrha oleogum resin essential oil were δ-elemene (2.0%), β-elemene (9.1%), γ-elemene (2.7%), germacrene D (3.3%), curzerene (29.7%), germacrene B (4.6%), furanoeudesma-1,3-diene (17.4%), and lindestrene (8.7%).
[Ulrich, Judith, et al. „Phytochemical Composition of Commiphora Oleogum Resins and Their Cytotoxicity against Skin Cancer Cells.“ Molecules 27.12 (2022): 3903] PDF (mdpi.com)
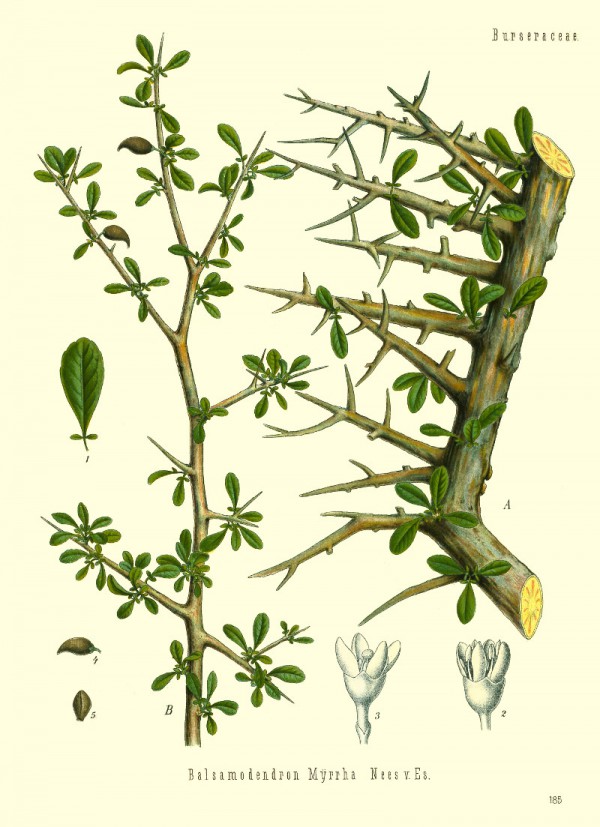
Köhler,F.E., Medizinal Pflanzen, vol.2 t.185 (1890)
http://plantgenera.org/species.php?id_species=267761