Arachis hypogaea L. - Fabaceae - peanut, Erdnuss
Annual herb, up to 70cm high, native to South America, also cultivated there and in India, Africa, China; leaves pinnate; flowers yellow, papilionaceous; fruit a pod (not a true nut), immature pods penetrate the soil and ripen underground, each pod contains 1-2(-6) outside reddish-brown seeds.
„Peanuts can be eaten raw, used in recipes, made into oils, textile materials, and peanut butter… used for snacks, confectionery and bakery products…“ wikipedia
Gas chromatography-olfactometry of headspace samples (GCOH) led to the identification of 21 odorants in peanut oil. Nine compounds were found in peanut, hazelnut, and pumpkin seed oils: Acetaldehyde, propanal, 2-methylpropanal, 2- and 3-methylbutanal, 2-ethyl-3,5-dimethylpyrazine, 2,3-diethyl-5-methylpyrazine, (E)-2-nonenal and vanillin. 1-Penten-3-one appeared alone with relative high FD-factor in peanut oil.
[A comparative study of potent odorants in peanut, hazelnut, and pumpkin seed oils on the basis of aroma extract dilution analysis (AEDA) and gas chromatography‐olfactometry of headspace samples (GCOH)., Matsui, T., Guth, H., & Grosch, W., Lipid/Fett, Vol.100(2), 1998, 51-56]
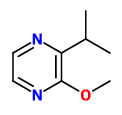
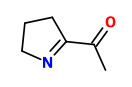
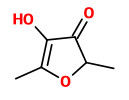
„Application of an aroma extract dilution analysis on an aroma distillate prepared from organically grown, raw West-African peanuts (Cameroon) revealed 36 odor-active areas in the flavor dilution (FD) factor range of 1 to 2048. The identification experiments, which were all performed by using the respective reference chemicals, revealed 2-isopropyl-3-methoxypyrazine (earthy, pea-like), 2-isobutyl-3-methoxypyrazine (bell pepper-like, earthy), and trans-4,5-epoxy-(E)-2-decenal (metallic) with the highest FD factors among the 36 aroma compounds identified. The two last mentioned odorants and another set of 22 further odorants were identified for the first time in raw peanuts. A comparative aroma extract dilution analysis applied on distillates prepared from either the raw peanuts or ground peanut meal roasted in a pan showed 52 odor-active areas in the FD factor range of 8 to 2048 in the roasted nut material. The identification experiments in combination with the FD factors revealed that among them, 2-acetyl-1-pyrroline and 4-hydroxy-2,5-dimethyl-3-(2H)-furanone showed the most significant contribution to the overall aroma, followed by 1-octen-3-one, 2-isopropyl-3-methoxypyrazine, (E,E)-2,4-decadienal, and trans-4,5-epoxy-(E)-2-decenal. As a further result, 20 aroma compounds were newly identified in roasted peanuts, such as 2-propionyl-1-pyrroline and 2-acetyltetrahydropyridine (both popcorn-like). In particular, 2-acetyl-1-pyrroline and 4-hydroxy-2,5-dimethyl-3(2H)-furanone (furaneol) showed the most pronounced increase after roasting.“
[Comparison of the key aroma compounds in organically grown, raw West-African peanuts (Arachis hypogaea) and in ground, pan-roasted meal produced thereof., Chetschik, I., Granvogl, M., Schieberle, P., Journal of agricultural and food chemistry, 56(21), 2008, 10237-10243]
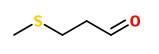
Of the 26 odor-active compounds (characterized by GC-olfactometry) present in raw peanuts, 3-isopropyl-2-methoxypyrazine (earthy beany), acetic acid, and 3-(methylthio)propanal (methional, musty potato tomato) showed the highest OAVs. Methanethiol, 2,3-pentanedione (buttery), 3-(methylthio)propanal, and 2- and 3-methylbutanal (aldehydic chocolate) as well as the intensely popcorn-like smelling 2-acetyl-1-pyrroline revealed the highest OAV of the 36 odor-active substances found in the pan-roasted peanut meal. Aroma recombination studies confirmed the importance, in particular, of methanethiol and of lipid degradation products in the characteristic aroma of the freshly roasted peanut material. Eight new found pyrazines did not show an impact on the overall aroma, as proved by adding them to a recombinate.
[Quantitation of key peanut aroma compounds in raw peanuts and pan-roasted peanut meal. Aroma reconstitution and comparison with commercial peanut products., Chetschik, I., Granvogl, M., Schieberle, P., Journal of agricultural and food chemistry, 58(20), 2010, 11018-11026]

Arachis hypogaea, Benin (2021) © ONG OeBenin CC BY-SA 4.0 inaturalist.org

Köhler,F.E., Medizinal Pflanzen, vol.3, t.42 (1890) plantgenera.org